
- P-ISSN 1225-0163
- E-ISSN 2288-8985

Test laboratories applying the requirements of ISO/IEC 17025 must estimate measurement uncertainty for quantitative measurements. Food analysis involves physical measurements and chemical analysis and a variety of principles and techniques are applied. The cause and magnitude of the uncertainty in both types of measurements can vary from measurement to measurement. In this review, measurement uncertainty is reinterpreted. The differences between measurement models and measurement functions, the relationship between fishbone and measurement models, and the application of bottom-up and top-down methods are compared, as well. In addition, the estimation of measurement uncertainty suitable for each analysis method in the physical and chemical analysis of food was compared and discussed. When the analytical process is complex, such as chemical analysis of food, it is considered a reasonable approach to adopt a top-down method to estimate measurement uncertainty by considering the overall uncertainty rather than decomposing the measurement result into its individual components.
Food analysis is essential to ensure the safety and quality of food consumed by humans and to promote a healthy diet by detecting harmful substances, pollutants, and allergens present in food and preventing their intake in advance. By analyzing the quantity and types of nutrients contained in a product, consumers can be equipped with information to make informed dietary choices.1 Food manufacturers can maintain and manage product quality through food analysis, thus providing consumers with the highest quality products by assessing factors such as freshness, degree of deterioration, and expiration date. Moreover, safety can be guaranteed by preventing the distribution of defective and adulterated food to consumers.
Food analysis is more complex than analysis in any other scientific field due to the variety of raw materials, the complexity of physical, chemical, and biological analytical methods, and the large number of analytical components. The analysis time is typically longer as well.2 Food analysis requires various analytical methods depending on the purpose of analysis, and the results can be applied in complex issues and disputes such as hygiene regulations, legal disputes, and trade. Therefore, more accurate results are demanded in food analysis, and interest in reliability is on the rise.3
The unification of the measurement system, decided at The Meter Convention held in 1875,4 brought about significant changes in the field of measurement. Various terms such as error, accuracy, precision, and uncertainty have since been used to indicate the reliability of measurement results. However, as expression methods vary depending on each measurement field, unified guidelines are gradually needed to indicate the reliability of measurement results. As awareness of the importance of measurement uncertainty spreads, the Bureau International des Poids et Mesures (BIPM) jointly created the Guide to the Expression of Uncertainty in Measurement (GUM) with international organizations related to metrology to unify the methods for evaluating and expressing measurement uncertainty.5 The guideline is intended to expand and apply the GUM approved by the International Committee of Weights and Measures (CIPM) in 1981 to industrial and commercial fields. Currently, it is widely adopted and applied because it is evaluated as the most appropriate approach to uncertainty assessment. The guideline was republished with the same content as an ISO/ IEC international document.6 The importance of measurement uncertainty is emphasized in ISO/IEC 17025 on general requirements for the competence of testing and calibration laboratories.7 In food analysis, both physical measurement and chemical analysis are included, employing different principles and techniques. Both types of measurements can be subject to uncertainty, and the causes and magnitudes may vary between measurements. The measurement uncertainty estimation method proposed by GUM is not sufficient to express measurement uncertainty in food analysis where different analytical methods are applied, such as chemical or instrumental analysis.
In this review, measurement uncertainty is reinterpreted, and the differences between measurement models and measurement functions, the relationship between the fishbone and measurement model, and the application of bottom-up and top-down methods are compared. The estimation of measurement uncertainty suitable for each analytical method in physical and chemical analysis of food is also compared and discussed.
The use of a measurement model in the prediction stage of measurement uncertainty is a method recommended by GUM. This guide explains how to estimate the uncertainty of the final measurement result by considering all factors that can occur during the measurement process.6 The most important and often error-producing factor in estimating measurement uncertainty is confusion between the measurement model and the measurement function. The measurement model described in GUM is a mathematical expression that predicts uncertainty by considering various factors and is used to estimate measurement uncertainty, including major factors that occur during the measurement process. The main advantages of using a measurement model are: First, the measurement model prevents the omission of all elements that occur during the measurement process, allowing for a more accurate prediction of the measurement uncertainty results. Second, it visually expresses measurement uncertainty by considering various factors in an integrated manner, enabling more accurate predictions than considering individual factors. Lastly, the measurement model can be applied to all general measurement processes. Therefore, uncertainty can be predicted consistently for multiple measurement processes. In measurement uncertainty, a measurement model is used to describe the relationship between the measured quantity and various sources of uncertainty that affect the measurement result, and is usually formulated Eq. (1):
h(Y, X1, …, Xn) = 0 (1)
Where
Y: The output quantity in the measurement model
X1, X2, ….. Xn : Input quantities
Where Y, the output quantity, is the measurand, the value of which is to be inferred from information about input quantities X1, …, Xn. In more complex cases where there are two or more output quantities, the measurement model consists of more than one equation. Therefore, the measurement model should be thought of as a set equations rather than an algebraic equations.
On the other hand, calculation formulas are used to determine the value of a measurand, which is the quantity being measured. This involves applying mathematical operations or algorithms to measured values or other related data. A calculation may include a formula, equation, or algorithm that converts a measured value into a desired quantity or provides information needed for further analysis or decision making. The focus is on obtaining the final result or value of the measurand rather than directly including the sum of the uncertainty contributions. In summary, measurement models are used to evaluate and quantify measurement uncertainty by considering the sum of uncertainty contributions, whereas measurement functions are used to calculate the measured value of a measurement object based on measured data without explicitly including the sum of uncertainty contributions. Thus, the measurement model for measurement uncertainty represents the total of measurement parameter uncertainties, making it a separate issue from the measurement function used to determine the quantity of the measurement object.
However, some scientists do not properly recognize the difference between the measurement model and the measurement function, and they easily make mistakes in estimating the measurement uncertainty. In the case of simple physical measurements, the factors of the measurement function can be directly applied to the measurement model. In complex and multi-stage chemical analyses, applying the measurement model only with the factors included in the measurement function will cause many errors. For example, a typical error in estimating the measurement uncertainty in moisture content analysis is applying the measurement function to the measurement model. The measurement function for moisture analysis is given by Eq. (2):8
Moisture content (%) = \(\begin{align}\frac{W S(g)-W D S(g)}{W S(g)} \times 100\end{align}\) (2)
Where
WS(g): Weight of wet sample
WDS(g): Weight of dried sample
If the measurement model is set to be the same as the measurement function, the measurement uncertainty is set as Eq. (3) below:9
Where
U: uncertainty of sample
u(linearity): linearity of balance
u(precision): precision of balance
The fishbone diagram is also set up as shown in the Fig. 1 by applying the terms of the measurement function, resulting in the error of estimating only the uncertainty of the balance from the measurement uncertainty of moisture.
The measurement model proposed by GUM is defined to include all factors that may cause uncertainty related to the measurement, so each uncertainty corresponding to all processes related to moisture measurement, such as grinding of the sample, laboratory sampling, drying conditions, uncertainty due to oven location, and repeat measurements, should be considered. For example, Tanaka included uncertainty factors such as, the mass of the test sample before drying, the mass of the sample after drying, the mass of the sample, the moisture dispersion due to temperature distribution in the dryer, the moisture dispersion due to the repeatability and variation between samples, and the moisture dispersion due to reproducibility of the grinder in the measurement model as in Eq. (4).10
Where
M: the moisture content of the lot of the sample (%)
m0: the mass of the sample before drying + the mass of the weighing can (g)
m1: the mass of the sample after drying + the mass of the weighing can (g)
mc: the mass of the weighing can (g)
εT: moisture dispersion caused by the temperature distribution in the dryer (%)
εSAM: moisture dispersion caused by deviation between the samples (%)
εRD: moisture dispersion caused by the repeatability of the drying process (%)
εRC: moisture dispersion caused by the repeatability of the crushed samples (%)
εG: moisture dispersion caused by the reproducibility of the grinders (%)
The fishbone diagram by Tanaka's measurement model in Fig. 2 appears to accurately include certain uncertainty factors that may occur in actual moisture analysis experiments when compared to the fishbone diagram by SINGLAS.
Whereas SINGLAS applied the mass of the sample before drying, the mass of the sample after drying, and the mass of the sample to the measurement model. Eventually only the uncertainty of the balance was calculated repeatedly.9 When the factors involved n estimating measurement results are diverse, uncertainty can be assessed using statistical methods, but, practical constraints such as time and resources often make this approach impractical. The measurement uncertainty results is typically evaluated using mathematical models of measurement and propagation laws of uncertainty. Implicit in this guide is the assumption that measurements can be mathematically modeled to meet the required level of accuracy. Measurement functions are crafted to derive the most accurate estimate of the quantity to be measured, drawing upon observed data and information from the employed measurement method. Typically, these functions involve mathematical operations like addition, subtraction, multiplication, division, and corrections or factors. However, they do not account for all uncertainty factors influencing the final uncertainty value. Therefore, estimating measurement uncertainty solely through the application of the measurement function is inadequate.
The measurement model is specifically tailored for estimating measurement uncertainty. As such, it should encompass factors not accounted for in the measurement function, incorporating all sources of uncertainty associated with the measurement process. This entails several elements, including pretreatment of the test sample such as collection, extraction, dilution, concentration, hydrolysis, derivatization, enzyme treatment, etc. Additionally, the measurement model should address uncertainty related to the measurement method itself, as well as statistical processing of the measurement results. A model serves as a representation of a system, process, concept, commonly employed to understand, analyze and predict behavior. It can manifest in various forms, including mathematical equations, diagrams, and computer simulations. Conversely, an equation constitutes a precise mathematical statement that asserts the equality of two expressions. Within the realm of modeling, equations are utilized to represent relationships between variables. Thus, while an equation offers a specific mathematical description, a model encompasses a broader concept that may incorporate equations as part of its expression. While a measurement function is utilized to ascertain the value of a measurement result, a measurement model is employed to estimate both the combined result and the standard uncertainty associated with it. So it is essential to distinguish between measurement function and measurement model when estimating measurement uncertainty.
In the process of estimating measurement uncertainty, once the measurement model is established, the fishbone diagram can be employed to identify and analyze potential sources of uncertainty or error that may arise during the measurement process based on the information. Fishbone diagrams assist in identifying potential sources of systematic or random errors, including equipment constraints, environmental conditions, calibration issues, and operator-related factors that can impact measurements and contribute to uncertainty.11 While measurement models offer a mathematical foundation for quantitatively estimating uncertainty, fishbone diagrams provide a more holistic and comprehensive approach to uncertainty estimation by visualizing and identifying various elements of uncertainty.12 The uncertainty factors outlined in the measurement model should ideally be explicitly addressed in the fishbone diagram. Measurement models and fishbone diagrams are meant to complement each other. However, many researchers are unaware that the uncertainty factors adopted in the measurement model must match those mentioned in the fishbone diagram. As depicted in Fig. 1 above, it is crucial to accurately account for these discrepancies, as they are often established in entirely different contexts.
Methods for analyzing food can be broadly categorized into physical, chemical and biological measurements. Different principles and techniques are applied to each field. Both physical and chemical measurements can be influenced by uncertainties related to measurement equipment limitations, environmental factors, human error, and other sources of variability. The sources and magnitude of uncertainty vary between physical and chemical measurements.13
Physical measurements involve the use of tools or equipment to observe or quantify the physical properties of an object or substance, such as length, mass, temperature, pressure, and volume.14 Uncertainty in physical measurements may arise from environmental factors such as temperature fluctuations, limitations of the measuring equipment used, and variations in gravity. Hence, precise conditions are essential during measurement.15 Once the test sample reaches equilibrium in a standard environment, it can be directly measured or observed without any pretreatment. Physical measurement is relatively straightforward and nondestructive compared to chemical measurement. In contrast, chemical measurement involves determining the chemical composition or properties of a test sample. This often includes destructive analysis, where the test sample is pulverized to extract and purify the measurement object, followed by the use of indicators or other chemical agents to detect or quantify the presence or concentration of specific chemical substances or components in the test sample. The analytical process for food may involve a variety of preprocessing steps, such as grinding, extraction, dilution, concentration, titration, hydrolysis, and derivatization, depending on the characteristics of the test sample being analyzed and the target components.16 Therefore, chemical measurements can be influenced by test sample preparation errors, chemical reactions, interferences, and other sources of variability related to the complexity of the measurement process, leading to various uncertainties.
The difference in measurement method can be likened to the act of taking a photo or drawing a picture of the same scene. Photography involves using a camera to capture an object, scene, or person by exposing a photosensitive material, such as film or a digital sensor, to light reflected or emitted from the object being photographed. The visual information is then processed and fixed on the photosensitive material or digitally recorded.17 On the other hand, painting is a visual art form expressed by applying pigments or other materials to surfaces such as canvas, paper, or wood. It is typically created by an artist who deliberately applies paint to a surface using a brush, palette knife, or other tools.18
When conducting physical measurements, the requirements for the measurement environment, such as pressure, temperature, and humidity in a standard room, are relatively simple but must be detailed and accurate. Similarly, when taking photos, the requirements for the measurement environment, such as illuminance, shutter speed, film sensitivity, and aperture, are relatively simple yet must be precisely controlled. In contrast, a painting studio does not require very strict conditions. Creating a painting is a complex process that demands a variety of tools, including different types of pigments, brushes, knives, canvas, and oil. Moreover, the outcome of a painting can vary significantly depending on the individual artist, introducing further differences.
As a result, in the case of physical measurements, it is possible to reproduce very similar products under the same measurement conditions. However, in paintings, even if the same materials and conditions are provided, it becomes quite difficult to reproduce similar products. This underscores the need for fundamentally different approaches to estimating measurement uncertainty for physical measurement and chemical analysis. So, the production process of photographs and paintings can be compared to the physical measurement and chemical analysis. Food analysis uses a variety of analytical methods depending on the measurement purpose, so the nature of the measurement method must first be considered to account for uncertainty in the measurement results. Just as there are clear differences in the properties of photography and painting, there are also differences in the properties of physical and chemical measurements. Therefore, estimation of measurement uncertainty must proceed through various paths depending on the measurement method. The choice between these methods may depend on factors such as the nature of the measurement process, data availability, resources, and time constraints.
ISO/GUM or known as bottom-up is widely regarded as one of the best approaches to estimating uncertainty. It allows the estimation of the measurement result to be expressed as a monomial expression, incorporating elements of uncertainty that may arise during the measurement process, so it can be applied as a measurement model. Initially, the measurement uncertainty was created based on the basic concepts of physical measurement. The results of all types of measurements should be presented with their associated uncertainties. GUM is the basic reference for uncertainty estimation applicable to all physical types of measurements such as dimensional, geometric, weight, volume, pressure, viscosity, current and temperature measurements. Uncertainty values indicate a lack of understanding of the measurement process. In other words, uncertainty is a quantification of doubt about a measurement. Factors affecting measurement uncertainty can be grouped into five categories: instrument, operator, workpiece, measurement procedure, and environment (e.g., ambient temperature and pressure). The GUM method stipulates that the uncertainty of the measurement result should be propagated from the uncertainty of each factor influencing the measurement. To propagate uncertainty, the GUM formula is defined as follows: Measurement results are expressed with a confidence interval determined from uncertainty, typically within the 95 % confidence interval. This uncertainty, calculated within the 95 % confidence interval, is referred to as extended uncertainty. In the context of physical measurements in food analysis, it is recommended to establish the measurement uncertainty based on the measurement model of GUM.
The bottom-up approach to measurement uncertainty estimation involves quantifying the individual sources of uncertainty in a measurement and then mathematically combining them to obtain an overall uncertainty estimate, and typically involves identifying all potential sources of uncertainty in the measurement process, such as instrument error, environmental conditions, calibration uncertainty, and other relevant factors. This method entails identifying and quantifying the contribution of each individual source of uncertainty in the measurement process and subsequently combining them using mathematical methods to produce a comprehensive assessment of the overall uncertainty of the measurement result. Each source of uncertainty is then identified through methods like calibration or repeatability studies, or alternatively characterized by estimating their size using techniques such as vendor specifications. The uncertainties estimated for each source are mathematically combined, usually using statistical methods like error propagation or uncertainty budgeting, to obtain an overall uncertainty estimate for the measurement result.
However, the bottom-up approach may have limitations, such as the assumption of independence of uncertainty, which may not always be true in practice. The correlation between various sources of uncertainty may need to be carefully considered, especially in complex measurement systems. Additionally, bottom-up approaches can be timeconsuming and resource-intensive as they may require extensive measurements and data analysis to accurately estimate the uncertainty of all relevant factors. Therefore, for physical measurements such as weight, size, and temperature of food, etc. it is safe to apply the bottom-up calculation method proposed in GUM.
In chemical analysis of food, measurement uncertainty can be estimated using the modeling approach proposed by GUM or the bottom-up method. All sources of uncertainty are identified, the contribution of each source is quantified, and then all contributions are combined into a budget to provide an estimate of the combined standard uncertainty. Previously, it was mentioned that physical measurement and chemical analysis were compared to the production process of photographs and paintings. In chemical analysis, instead of employing a modeling approach that lists and calculates all factors individually, as with the measurement uncertainty measurement model proposed by GUM, the analysis process is subdivided considering that it consists of each unit operation. Detailed procedures must then be established based on this subdivision.
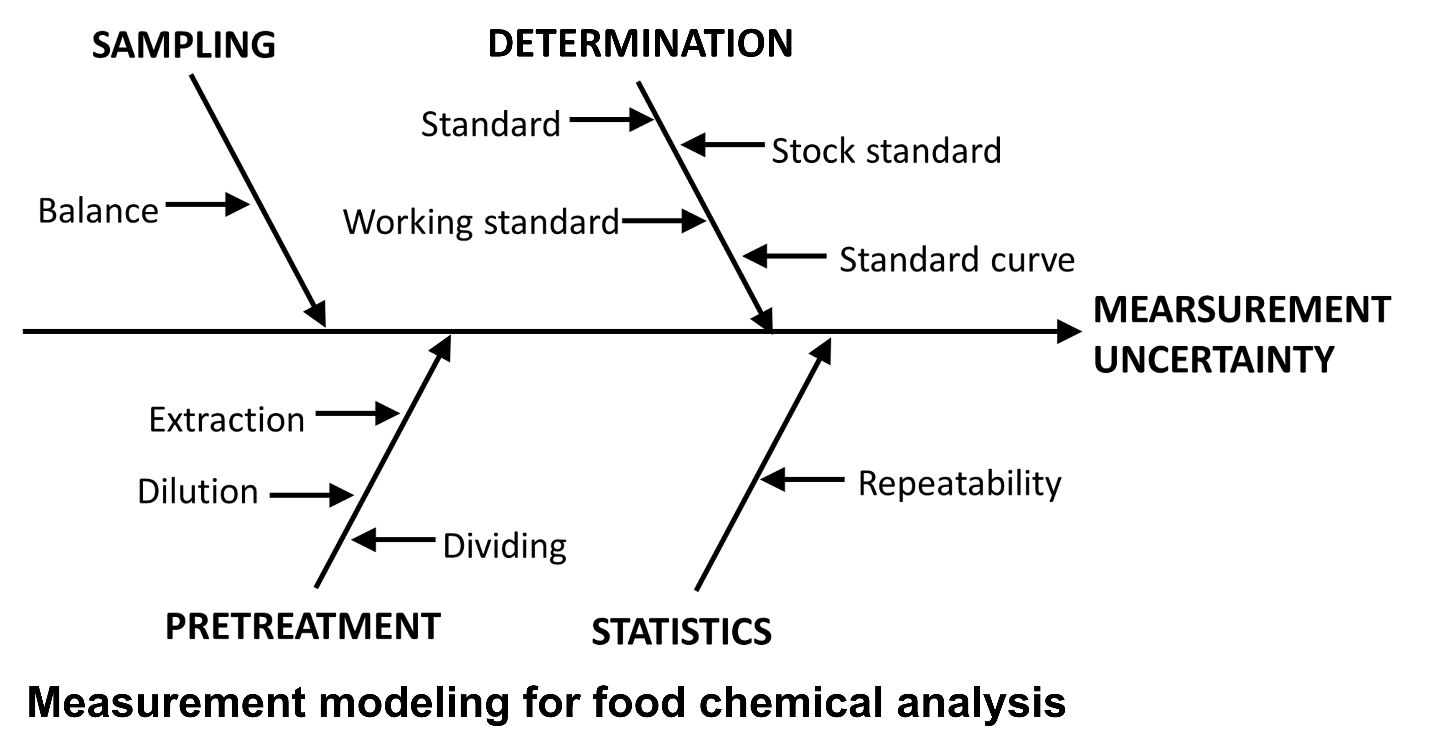
In Quantifying Uncertainty in Analytical Measurement (QUAM),19 the measurement uncertainty estimation process in general chemical analysis can be broadly divided into four steps, as shown in Fig. 2. Consequently, there is no need to create a complicated fishbone diagram; instead, one can simply set up a large stem and add details of the measurement actions performed at each step. If organized according to unit operation, the process in chemical analysis can be broadly divided into four stages: sampling, pretreatment, analysis, and statistical processing.20 Therefore, when establishing a measurement model for measurement uncertainty, the most systematic way suggested to formulate, is to sum up the uncertainties of these steps (Eq. (5)).
Y = f(Xsampling, Xpretreatment, Xdetermination, Xstatistics) (5)
Where
Y: measurement uncertainty
Xsampling: uncertainty in sampling process
Xpretreatment: uncertainty in pretreatment process
Xdetermination: uncertainty in determination process
Xstatistics: uncertainty in statistics process
This can be expressed as a fishbone diagram as follows.
First, in sampling, the uncertainty of the balance is estimated, and then in the pretreatment process, the uncertainty for the volumetric flask, pipette, etc., used in the test sample extraction, dilution, division, and recovery rate determination shall be estimated. In the subsequent analysis (measurement) stage, it is considered appropriate to estimate the uncertainty for chemical standards, stock solutions, working standards, calibration curves, and device sensitivities. Finally, the uncertainty obtained from statistical processing based on repeated measurements could be provided.
After the publication of the ISO/GUM guide, the QUAM guide was subsequently published by EURACHEM/CITAC.19 This guide contains useful procedures for estimating measurement uncertainty in analytical measurements. However, this approach is deemed tedious, time-consuming, and impractical from an analytical standpoint due to its inability to accommodate all the current procedures and principles applied in analytical chemistry, particularly concerning matrix effects, sampling operations, and interferences.21,22
In wet chemical analysis or instrumental analysis using GC or HPLC, estimating uncertainty using bottom-up methods can be somewhat challenging. Unlike physical measurements, which often have relatively simple and standardized measurement formats, chemical analysis necessitates varying environments in each laboratory during the extraction, pretreatment, and measurement of the analyte. Additionally, in chemical analysis, the array of pretreatment devices and instruments used is diverse and complex, making it challenging to individually estimate the uncertainty for all measurement processes, especially due to the influence of the experimenter's skill level. For this reason, the top-down method is often favored over the bottom-up method in chemical analysis. Topdown measurement uncertainty estimation is often utilized in situations where accurately determining individual sources of uncertainty is difficult or impractical, or when the focus is on understanding the overall uncertainty of the measurement system rather than the detailed contribution of each uncertainty source. Besides the Eurachem/CITAC guide, the topdown approach has been systematically detailed in several documents, including the Eurolab report on alternative approaches to uncertainty assessment and ISO 21748.19,23-25 Guidelines published by the ISO and Clinical and Laboratory Standards Institute (CLSI) recommend a top-down approach for measurement uncertainty estimation in clinical laboratories.26-28
Pan Liu addressed the limitations of the classic uncertainty assessment method in quality control, such as repeated evaluation, omission, and difficulty in quantifying uncertainty components. They shifted focus to the top-down gray system theory, exemplified by the control chart method, and proposed a new evaluation method for measurement uncertainty.29 Flávia Martinello et al. asserted that top-down approaches typically directly estimate measurement uncertainty by evaluating quality control data or method validation experiment data, emphasizing their practicality, cost-effectiveness, and routine applicability. They noted that these approaches could be regularly updated with additional data from Internal Quality Control and proficiency testing results.30 Carla Palma et al. also emphasized that uncertainty serves as a scientific indicator of the quality level and variance of measured results, taking into account the shortcomings of the current classical uncertainty assessment method (i.e., GUM method) such as repeated evaluation, omission, and difficulty in quantifying uncertainty components. They proposed a top-down approach represented by the projection method.31 According to SAC, adopting a holistic, top-down approach to assessing measurement uncertainty in chemical and microbiological analyses entails a much simpler evaluation process and offers dynamic or current uncertainty levels in reported test results. In contrast, the traditional ISO GUM (bottom-up) method was described as tedious and complex in its evaluation.9 Lee et al. compared measurement uncertainty using two approaches, bottom-up and top-down, and found that the uncertainty of the bottom-up approach was very similar to that of the top-down approach. This demonstrates that the two approaches are almost identical and interchangeable. The uncertainties at the low glucose concentration by the bottom-up and top-down were ±0.18 mmol/L and ±0.17 mmol/L, respectively (all k = 2). Those at the high glucose concentration by the bottom-up and top-down were ±0.34 mmol/L and ±0.36 mmol/L, respectively (all k = 2). The clinical laboratory concluded that measurement uncertainty can be determined with a simpler top-down approach.32 Medina-Pastor et al. suggested that a top-down approach is best suited for assessing measurement uncertainty in pesticide residue testing laboratories. They found that the opposite approach, bottom-up assessment of measurement uncertainty, leads to significant difficulties in consistently assessing all pesticides.33 Additionally, they introduced in several other literatures that the top-down approach is an economical and reasonable method of estimating uncertainty in food chemical measurements compared to the classic bottom-up approach.19
The definition of measurement uncertainty is well articulated in GUM and VIM. However, these expressions are often deemed too abstract. To provide a more scientific description, measurement uncertainty can be considered as the aggregate of uncertainties associated with the tools used for measurement. When estimating uncertainty, a measurement model and fishbone diagram are applied to prevent the omission of uncertainty factors and to identify their respective contributions. The arguments presented in these two methods are selected in accordance with the same definition and are nearly identical. There are several approaches that can be used to estimate the uncertainty of a measurement result, and it is important that whichever approach is used, it is scientifically acceptable. Neither of these scientifically accepted approaches can be said to be better than the other. In other words, there is no hierarchy in this approach. Choosing an appropriate approach will depend on the type of measurement or analysis, the method used, the level of reliability required, and the urgency of the request to estimate measurement uncertainty. Typically, procedures are based on either a bottom-up or top-down approach, with the latter utilizing data from collaborative studies, proficiency studies, validation studies, in-laboratory quality control samples, or a combination. In most physical measurement fields, the measurement uncertainty measurement model and measurement function may be the same according to the GUM method. In chemical analysis, the unit operation of the measurement process must be identified, and the measurement uncertainty for each operation must be calculated. Therefore, the measurement model and measurement function cannot be the same. In recent years, measurement uncertainty in chemical analysis has increasingly been estimated using the top-down method. This approach estimates the measurement uncertainty by considering the overall uncertainty without decomposing the measurement result into individual components. Top down process is persuasive for estimating the measurement uncertainty of chemical analysis, in cases where the analysis process is complex.