Introduction
Sea-level rise and the preservation of coastal ecosystems in Korea
Coastal ecosystems—such as wetlands, marshes, mudflats, reclaimed lands, and castal sand dunes—are biodiversity hotspots shaped by long-standing interactions between coastal communities and nature. These habitats are not only ecological reservoirs but also cultural landscapes that reflect long-standing interactions between human communities and coastal nature. However, their integrity is increasingly jeopardized—not solely by sea-level rise, but also by associated changes in seawater temperature and salinity. Ectothermic marine organisms, including many fish and invertebrates, are particularly sensitive to such changes (Noto & Shurin, 2017).
In recent decades, the climate crisis and sea-level rise have emerged not only as pressing environmental issues but also as profound challenges to the ecological fabric of coastal societies. Coastal ecosystems, home to specialized plant species such as halophytes, play a vital role in maintaining ecological balance, supporting biodiversity, and sustaining cultural traditions deeply tied to the land and sea. Halophytes—plants adapted to high salinity—thrive in salt marshes and tidal flats, habitats that have shaped and been shaped by centuries of human interaction, local knowledge, and sustainable practices (Castillo & Moreno-Casasola, 1996; Ihm et al., 2007).
From an ecological standpoint, sea-level rise threatens not only biodiversity but also the interwoven systems of life, knowledge, and tradition. As rising seas submerge low-lying coastal areas, halophytes face habitat loss and altered salinity regimes, which may challenge their survival and disrupt the complex ecological functions they support (Janousek et al., 2016; Ward et al., 2016). These changes affect not only natural processes but also the cultural continuity of coastal communities that depend on these ecosystems for food, protection, and identity.
Historically, global sea levels have been rising gradually, with significant acceleration in the 20th century due to industrialization. The global average rate has increased from 1.7 mm/year in the early 1900s to 1.8 mm/year between 1961 and 2003 (Hong et al., 2010). The key drivers include thermal expansion, melting glaciers, and anthropogenic influences on freshwater systems (Alahuhta et al., 2011). In Korea, the impact has been more pronounced: sea levels are rising at an average rate of 4.02 mm/year, exceeding the global average and suggesting heightened regional vulnerability (Kim, 2012). This places considerable stress on coastal habitats in the Yellow Sea, East Sea, and South Sea.
Wetlands, which serve as critical buffers against storm surges and carbon sinks, are disappearing at an alarming rate globally—over half of such areas have been lost in countries like Myanmar and Malaysia (Boyle & Ardill, 1989). Their loss represents not only an ecological disaster but also the erosion of traditional ecological knowledge and the cultural heritage embedded in these landscapes. The displacement of plant and animal species is compounded by modern infrastructure, which restricts natural migration paths and hinders ecological resilience.
Within this broader context, halophytes become more than just botanical subjects—they emerge as key ecological actors. Their capacity to adapt, stabilize shorelines, and sequester carbon underscores their significance in both ecosystem conservation and climate adaptation strategies (Forey et al., 2008; Myeong, 2010). Moreover, the inland migration of halophytes, where possible, may create hybrid ecotones—new interfaces between terrestrial and saline ecosystems—offering opportunities for renewed ecological interaction.
This study investigates how salt marsh plants in South Korea respond strategically to sea level-induced variations in canopy height, using the Competitor, Stress-tolerator, Ruderal (CSR) framework (Hodgson et al., 1999; Pierce et al., 2013). By analyzing shifts in plant strategies, we aim to deepen our understanding of how ecological responses can inform broader conservation efforts rooted in both science and cultural stewardship.
Materials and Methods
Site description and plant survey
This study focuses on three halophyte species—Salicornia europaea, Suaeda glauca, and Suaeda maritima—inhabiting the salt marshes along the Haeun-ri, Hyeongyeong-myeon, Muan-gun coastline in South Korea, a region increasingly affected by sea-level rise. Plant communities were surveyed both in mainland coastal zones of Muan-gun and the nearby islands of Shinan-gun (Hyun et al., 2018; Jang et al., 2014; 2021; 2022; Kim, 1984; 1986; Lee et al., 2020; Son et al., 2020; Yang et al., 2021). These salt marshes are ecologically significant, serving as critical interfaces between land and sea, and supporting biodiversity, traditional fisheries and octopus fishing, and the livelihoods of local communities. The plant samples were collected with a conservation-oriented approach, minimizing disturbance to native habitats and respecting the ecological functions of these landscapes.
Analysis of plant morphological factors
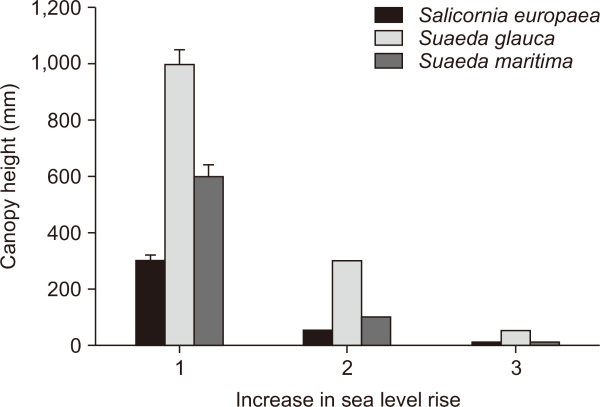
To assess the adaptive responses of native halophytes to environmental change, we evaluated morphological and ecological characteristics of each species (Hodgson et al., 1999), including diversity, coverage, and density (Lee et al., 2020). Additional quantitative variables related to plant form and leaf traits were measured (Pierce et al., 2007; 2013): canopy height (mm), lateral spread (mm), leaf area (mm2), leaf dry weight (mg), leaf dry matter content (%), specific leaf area (mm2 mg–1), flowering period, and flowering start. Canopy height, a critical indicator in plant ecological strategy, was hypothesized to respond dynamically to sea-level rise and tidal influences (Janousek et al., 2016; Ward et al., 2016) (Fig. 1). The remaining traits were assumed to be relatively stable across tidal conditions. These measurements inform not only plant functional responses but also contribute to broader conservation planning by identifying resilient species under coastal stress.
Competitor, Stress-tolerator, Ruderal analysis
CSR strategies were calculated and visualized in a ternary diagram using the methodology proposed by Grime (1974) and Hodgson et al. (1999) (Tables 1Table 2-3, Fig. 1). The analysis aimed to explore how plant ecological strategies shift in response to rising sea levels.
Species characteristics
S. europaea is commonly found in saltmarshes and saline coastal habitats, where it serves as a pioneer species. Its role in initiating ecological succession and stabilizing intertidal zones is fundamental to both habitat conservation and shoreline protection (Cho, 2006; Lefor et al., 1987; Shim et al., 2009; Taiz & Zeiger, 2006).
S. glauca typically inhabits saline mudflats and coastal margins. With its thick, water-storing leaves and specialized salt glands for excreting excess sodium, this species demonstrates key adaptations that enable survival under high salinity.
Similarly, S. maritima is a vital component of saltmarsh ecosystems, thriving under fluctuating salinity, tidal variation, and nutrient stress. Its presence enhances ecosystem resilience by initiating vegetation development in saline environments, ultimately supporting the formation of more complex plant communities.
Results
A strong linear relationship was observed between sea-level rise and changes in halophyte CSR strategies, particularly in the competitive (C) and ruderal (R) components (Fig. 2).
• C strategy: Y=48.79–0.0286X (r=0.91, P<0.001)
• R strategy: Y=51.21+0.0286X (r=0.91, P<0.001)
In the S. europaea community, the CSR classification shifted from R/CR (31:0:69) at a canopy height of 300 mm, to R (19:0:81) at 50 mm, and remained R (15:0:85) at 10 mm (Hodgson et al.,1999). (Table 3, Fig. 3).
For S. glauca, the strategy changed from CR (44:0:56) at 1,000 mm to R (30:0:70) at 300 mm, and R (18:0:82) at 50 mm.
In the case of S. maritima, the CSR strategy shifted from CR (43:0:57) at 600 mm to R/CR (30:0:70) at 100 mm and remained R/CR (21:0:79) at 10 mm.
The strategic transitions observed were as follows:
• S. europaea: R/CR→R→R
• S. glauca: CR→R→R
• S. maritima: CR→R→R (Fig. 3).
Discussion
Vegetation, a key structural component of coastal ecosystems, is highly susceptible to changing hydrodynamic conditions (DeLaune et al., 1987; Sharpe & Baldwin, 2012). In salt marshes, plant species have been categorized according to their ecological strategies—such as CR, C, R/CR, and SC—based on their responses to competition, stress, and disturbance (Lee & Kim, 2018; Son et al., 2020). For instance, Artemisia scoparia, S. maritima, and Limonium tetragonum exhibit CR strategies, indicating adaptive capacities in fluctuating saline environments. Lateral root growth and nutrient storage, as observed in these taxa, signify evolutionary resilience to disturbance (Janousek et al., 2016).
In the study area, S. europaea and S. glauca both exhibited R/CR or CR strategies under conditions of increased tidal stress (Table 3, Fig. 3). As sea levels rose, the proportion of competitive strategies declined while ruderal strategies increased, indicating a shift in ecological function and resilience mechanisms. Notably, species classified as R/CR were annuals with an average canopy height of 269 mm, indicating a functional response that balances disturbance tolerance with rapid regrowth—traits critical to the persistence of coastal vegetation.
These findings contribute to a broader ecological understanding of halophyte communities. As key elements in South Korea’s coastal heritage and ecological networks, these plants serve not only biological functions—such as sediment trapping and shoreline protection—but also cultural roles in local traditions and ecological knowledge systems. Preserving these adaptive species and their habitats is therefore essential not only for biodiversity conservation but also for sustaining the cultural landscapes and livelihoods that depend on them.
The dominance of R and CR strategies in coastal zones indicates niche differentiation under high disturbance regimes. These plants, often short-lived but fast-reproducing, exemplify adaptation to fluctuating resources and harsh abiotic stress (Janousek et al., 2016; Pierce et al., 2007). From an ecological perspective, such species also embody traditional knowledge—used in food or land management—thus linking biodiversity with intangible cultural heritage.
On coastal dunes, 60% of species demonstrated CR/CSR strategies (Costa et al., 1996; Elmas, 2017), including Carex kobomugi, Ipomoea imperati, and S. europaea. An additional 20% exhibited SC/CSR strategies. Human disturbances, such as trampling and construction, favor ruderal species and accelerate ecological succession toward opportunistic plant communities (Ciccarelli, 2015). Understanding these biotic shifts is critical not only for biodiversity maintenance but also for the protection of culturally significant coastal landscapes.
Reclaimed lands represent socio-ecological frontiers where anthropogenic and natural processes intersect. Here, R or CR-dominant strategies (e.g., R/CR, CR) are common, as seen in species like Echinochloa crus-galli, Portulaca oleracea, Soncous brachyotus, and Setaria viridis (Min & Kim, 1999a; 1999b; Yang et al., 2021). Successional trends reveal a gradual transition from ruderal to competitive strategies, highlighting ecosystem maturation. These findings reflect not only ecological processes but also the cultural narrative of land use and reclamation, particularly along Korea’s west coast (Lee et al., 2020).
Simulations of sea-level rise revealed dynamic changes in CSR strategies. In S. europaea communities, a shift from R/CR to R was observed with reduced canopy height. Similarly, S. glauca and S. maritima communities displayed transitions from CR to R. These strategic shifts, driven by environmental stress, underline the fragility of plant community structures under climate change and highlight the importance of early intervention. These shifts signify a transition toward ruderal dominance in response to increasing environmental disturbance. Ruderal strategies are typically associated with short life cycles, rapid reproduction, and tolerance to unstable conditions—traits advantageous under rising sea levels and increased tidal frequency. Importantly, these adaptations support the regeneration capacity and soil stabilization roles of saltmarshes, which are vital for maintaining ecosystem integrity.
To sustain halophyte competitiveness amid rising seas, conservation strategies must be ecologically grounded. First, restoring degraded salt marshes enhances ecosystem resilience. Managed retreat and the facilitation of community migration may be appropriate in highly vulnerable areas. Elevating marsh surfaces through sediment augmentation or natural deposition processes can mitigate submergence risks. Riverine sediment supply must be maintained to support dynamic habitat renewal.
Despite growing concern, our understanding of how natural ecosystems respond to rapid climate shifts remains limited (Boyle & Ardill, 1989; Mueller et al., 2020; Reed, 1995; Ward et al., 2016). This uncertainty hinders accurate forecasting and proactive conservation planning. The compound effects of sea-level rise, pollution, and resource exploitation may produce synergetic damages greater than the sum of their parts. A precautionary and culturally informed conservation approach is therefore essential.
Safeguarding genetic resources is equally vital. Native halophyte seeds should be systematically collected and stored in seed banks, ensuring long-term biodiversity preservation. Community-based propagation and restoration programs can engage local stakeholders, strengthening ecological stewardship. Ultimately, integrating simple trait-based monitoring with traditional ecological knowledge offers a robust framework for future conservation of Korea’s southwest coastal ecosystems.
Author Contributions
Conceptualization: JWK, SYK. Data curation: MJK. Formal analysis: JC. Funding acquisition: JWK. Investigation: SYK. Supervision: JWK. Visualization: JC. Writing – original draft: SYK, JWK. Writing – review & editing: SYK, JWK.
Funding
This Research was supported by Research Funds of Mokpo National University in 2024.
Figures and Tables
Fig. 2
Relationship between sea level rise and competitive and ruderal strategies (%) of halophyte traits of the southwestern coast in South Korea. C strategy: Y=48.79–0.0286X (r=0.91, P<0.001); R strategy: Y=51.21+0.0286X (r=0.91, P<0.001). C, Competitor; S, Stress-tolerator; R, Rederal.
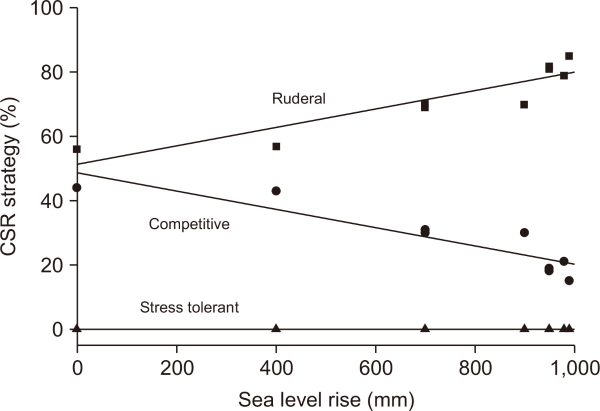
Fig. 3
Change of their CSR strategies from Hodgson et al. (1999) related to sea-level rise of halophyte traits of the southwestern coast in South Korea. (A) Salicornia europaea, (B) Suaeda glauca and (C) Suaeda maritima. Source: Hodgson et al. (1999). C, Competitor; S, Stress-tolerator; R, Rederal.
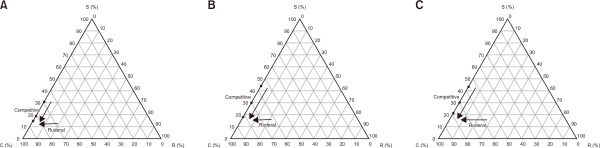
Table 1
Definitions of the predictor variables used the CSR allocation procedure
| Variable | Definition | ||
|---|---|---|---|
| Canopy height | Six-point classification | 1 | 1-49 mm |
| 2 | 50-99 mm | ||
| 3 | 100-299 mm | ||
| 4 | 300-599 mm | ||
| 5 | 600-999 mm | ||
| 6 | >999 mm | ||
| Dry matter content | Mean of percent dry matter content in the largest, fully hydrated, fully expanded leaves (%) | ||
| Flowering period | Normal duration of flowering period (mo) | ||
| Flowering start | Six-point classification | 1 | First flowering in March or earlier |
| 2 | in April | ||
| 3 | in May | ||
| 4 | in June | ||
| 5 | in July | ||
| 6 | in August or later, or before leaves in spring | ||
| Lateral spread | Six-point classification | 1 | 1: Plant short-lived |
| 2 | Loose tufted ramets radiating about a single axis, no thickened rootstock (in graminoids) | ||
| 2 | Compactly tufted about a single axis, no thickened rootstock (in non-graminoids) | ||
| 3 | Compactly tufted ramets appressed to each other at base (in graminoids) | ||
| 3 | Compactly tufted about a single axis, thickened rootstock present (in non-graminoids) | ||
| 4 | Shortly creeping, <40 mm between ramets | ||
| 5 | Creeping, 40-79 mm between ramets | ||
| 6 | Widely creeping, >79 mm between ramets | ||
| Leaf dry weight | Natural logarithm of mean dry weight in the largest, fully hydrated, fully expanded leaves (mg), plus 3 | ||
| Specific leaf area | Mean of area/dry weight quotient in the largest, hydrated, fully expanded leaves (mm²/mg) | ||
Table 2
Mean statistics related to sea-level rise of halophyte plant traits
| Properties | Canopy height (mm) |
Leaf dry matter content (%) |
Flowering period (# of mo) |
Lateral spread (six-point model) | Leaf dry weight (mg) |
Specific leaf area (mm2 mg–1) |
Flowering start (mo) |
|---|---|---|---|---|---|---|---|
| Average | 269 | 9 | 2 | 1 | 70 | 13 | 6 |
| N | 9 | 9 | 9 | 9 | 9 | 9 | 9 |
| Standard deviation | 336 | 1 | 1 | 1 | 30 | 2 | 1 |
| Max | 1,000 | 10 | 3 | 2 | 110 | 16 | 6 |
| Min | 10 | 9 | 2 | 1 | 47 | 12 | 5 |
Table 3
Plant species, their C (%), S (%), R (%) percentages, and CSR strategies from Hodgson et al. (1999) related to sea-level rise of halophyte plant traits of the southwestern coast in Korea
| Dominant community | Sea-level rise | % from Hodgson et al. (1999) | Strategy type | ||
|---|---|---|---|---|---|
|
|
|||||
| C (%) | S (%) | R (%) | |||
| Salicornia europaea | 300 | 31 | 0 | 69 | R/CR |
| 50 | 19 | 0 | 81 | R | |
| 10 | 15 | 0 | 85 | R | |
| Suaeda glauca | 1,000 | 44 | 0 | 56 | CR |
| 300 | 30 | 0 | 70 | R | |
| 50 | 18 | 0 | 82 | R | |
| Suaeda maritima | 600 | 43 | 0 | 57 | CR |
| 100 | 30 | 0 | 70 | R | |
| 10 | 21 | 0 | 79 | R | |