Introduction
Marine invertebrates are well known for their complicated lifecycles (Carr et al., 2017; Chen, 2021; Fuchs et al., 2020; Pandori & Sorte, 2019). They play an important role in ecosystem function and provide various ecosystem services to humans (Alves et al., 2020; Beseres Pollack et al., 2013; Chen, 2021; Spalding et al., 2017). Despite the growing number of studies on marine invertebrates, a substantial gap remains in our understanding, particularly regarding their tolerance to changing abiotic conditions (e.g., wave impact and desiccation) (Bini et al., 2006; Cardoso et al., 2011; Chen, 2021; Hortal et al., 2015).
In marine ecosystems, the settlement of invertebrate larvae is a unique behavior that ends the buoyant pelagic stage and begins the sedentary benthic stage (McClary, 1997). In general, this unique behavior tends to occur before metamorphosis (morphological and physiological changes from pelagic stage to benthic stage), with some exceptions (Scheltema, 1974). This behavior is influenced by various hydrodynamic factors, including the visual cues of substrates, surface characteristics, gravity, and chemicals (McClary, 1997). Previous studies have suggested that although larvae respond to flow conditions first, which causes them to swim to the bottom and explore, these are the chemical cues that actually induce settlement and metamorphosis (Rittschof et al., 1984; 1985; Zimme-Faust & Tamburri, 1994). To date, many studies have been performed on marine invertebrates to determine the key hydrodynamic factors for settlement and metamorphosis (McClary, 1997). Furthermore, phenotypic plasticity in marine organisms has recently been used as a bioindicator to quantify the relative hydrodynamic differences among locations (Reustle et al., 2023). However, little is known about why invertebrate species (e.g., barnacles) exhibit permanent settlement patterns (e.g., settlement angles against water currents) in benthic adulthood (Crisp & Stubbings, 1957). Such natural mechanisms can be applied to the development of innovative technologies in the field of biomimicry, such as design and architecture.
Barnacles are invertebrate species found in marine ecosystems worldwide and adhere strongly to rocky substrates (Shaw et al., 2024). Balanoid barnacles are sessile crustaceans with a conical, volcano-like shape surrounded by a hard, calcareous shell made up of a series of parietal plates sitting directly on a basal plate or membrane (Lively, 1986). Opercular plates (n=6) articulate at the upper portion of parietal plates, allowing feeding organs to emerge (Chan et al., 2021; Pérez-Losada et al., 2014). These species are greatly affected by hydrodynamics and therefore tend to cope with notable shifts in flow regimes from birth to death (Reustle et al., 2023). They respond behaviorally to changes in flow direction instantaneously and even appear to anticipate oscillating flow (Trager et al., 1990). To date, studies on the settlement angles of barnacle species have rarely been conducted. Therefore, further studies on the shell orientations of barnacles are required to understand their ecology.
In the present study, we examined the shell orientation of barnacles in response to wave forces in an artificial rocky intertidal zone. In addition, we compared the differences in shell orientation between two barnacle species of different sizes and shapes.
Materials and Methods
Sampling sites and samples
This study site is located in an artificial rocky intertidal zone of Daejang Island of Gunsan-si, Jeollabuk-do, South Korea (35° 50’ N, 126° 29’ E; Fig. 1). Barnacles were sampled using a digital camera, Nikon D6 (Nikon Corp., Tokyo, Japan) along the high-intertidal zone where two barnacle species (Fistulobalanus albicostatus and Chthamalus challengeri) coexisted along the coastline of Daejang Island under severe desiccation conditions (Fig. 2). Two barnacle species were observed and analyzed as target species in this study. Sampling was conducted at noon in the spring, on March 14th, 2025. The temperature of the study site was around 8°C under weak wind.
Data collections and statistical analyses
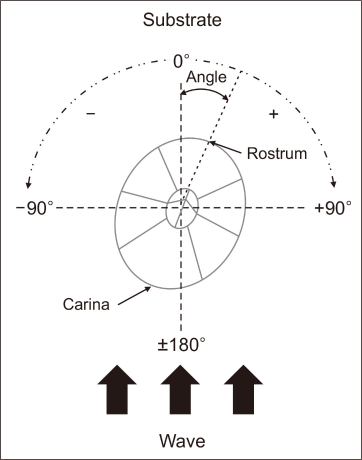
Photographs were taken to measure the percent covers (%), densities (number of individuals/cm2), sizes (length and width of shells; mm), and settlement orientations (angles to wave; °). Five quadrats (30×30 cm) were placed on an artificial concrete structure (vertical wall). Each plot was systematically placed along the vertical wall with a 50 cm gap. The percent cover, density, size, and shell orientation of the barnacles were visually estimated on a computer monitor using ImangeJ software version 1.54 g (National Institute of Mental Health, Bethesda, MD, USA). Five plots were analyzed to measure the percent cover of barnacles (Fig. 3). However, only one representative subplot (3×3 cm) for each plot was used to measure the density of barnacles from a total of five subplots (Fig. 4). We also measured the size (shell length and width) of barnacles randomly selected from two species (n=10 from each plot, totaling 50 inds. for each species). The ratio between length and width was calculated (Fig. 5). Finally, we measured the shell orientation across the wave (0°<vertical angle<+180° for right side or –180°<vertical angle<0° for left side) based on the rostrum pointing upstream and carina pointing downstream (Figs. 6, 7). The same barnacle samples were used to measure size and orientation. All measurements were analyzed using parametric one-way analysis of variance (ANOVA) because there was no violation of normality and homogeneity of variance (Table 1). Settlement orientation was analyzed using a non-parametric Mann-Whitney U-test due to the unequal variance between the two barnacle groups. All statistical analyses were performed using Jamovi version 2.2.5.5 (Mettler Toledo, Columbus, OH, USA).
Results
Percent cover and density
The percent cover (n=5 plots) and density (n=5 subplots) of each barnacle species are presented as box plots in Figs. 3, 4, respectively. The percent cover (mean±standard deviation [SD]) of F. albicostatus (49.4±17.2%) was very similar to that of C. challengeri (50.6±17.2%) in the rocky intertidal zone (P=0.915; Table 1). However, the density (mean±SD) of F. albicostatus (0.6±0.1 inds./cm2) was significantly lower than C. challengeri (2.8±1.2 inds./cm2) (P<0.050; Table 1).
Size
Sizes (mean±SD) of the two barnacle species (n=50 inds., 10 for each plot) are presented in Fig. 5. F. albicostatus (9.3±1.5 mm) showed a significantly higher length of shell than C. challengeri (4.8±0.8 mm) (Fig. 5A). Likewise, F. albicostatus (8.6±1.5 mm) showed a significantly higher width of shell than C. challengeri (4.0±0.7 mm) (Fig. 5B). However, the ratio of length/width showed that C. challengeri (1.2±0.2) was significantly higher than that of F. albicostatus (1.1±0.1) (Fig. 5C). The statistical results of all measurements using one-way ANOVA showed P<0.001 between the two barnacle species (Table 1). We could not measure the heights of the shells of the two species in the photographs. However, F. albicostatus, with a long conical shape, was clearly taller than C. challengeri, which has a short, flat shape (Fig. 2).
Settlement orientation
The shell orientations of the barnacles (n=50 inds., 10 for each plot) are presented in Fig. 7 as final parameters. We used the same individuals selected for the morphological measurements and measured their shell angles based on the rostrum pointing upstream and the carina pointing downstream (Fig. 6). Consequently, the settlement angles of the shells of the two barnacle species were small, approximately 10°. However, there was no significant difference between F. albicostatus (−2.34±29.0°) and C. challengeri (−9.30±39.8°) (Mann-Whitney U-test, P=0.287; Table 1).
Discussion
As one of the common arthropods in wave-swept marine environments, barnacles exhibit notable phenotypic plasticity in the dimensions of their feeding and reproductive organs in response to flow velocity (Reustle et al., 2023). However, barnacle cyprid larvae sometimes tend to settle in cracks and pits because they have low flow velocities or promote stronger antennular adhesion (McClary, 1997). Therefore, it remains unclear whether cyprids respond to hydrodynamics and/or surface texture (Mullineaux & Butman, 1991). Rittschof et al. (1984) showed that cyprids of the barnacle Balanus amphitrite were unaffected by flow during their initial contact with a surface with constant topography but were affected by the flow direction during exploration and attachment. This result suggests that the flow direction might affect the success of settlement on surfaces with constant topography in rocky intertidal zones.
To date, the effect of flow direction on barnacles after settlement has rarely been studied. However, barnacles generally tend to settle at an appropriate angle against the vertical tidal waves. Moore (1933) measured the orientation of young and older individuals of Balanus balanoides and interpreted the difference that he found to be due to the rotation of the shell during growth. In addition, Crisp (1953) showed that in certain species with canals in the basis, a change in the orientation of the shell often accompanied growth, and confirmed Moore’s view (1935) that the direction of rotation corresponded with that of the twist of the basal canals. Crisp and Stubbings (1957) reported an exceptional case of adult barnacle orientation. According to their study, barnacles (B. balanoides) tend to have shell orientations, with the rostrum pointing upstream and the carina pointing downstream. As a result of their study, the two different species of barnacles all showed small settlement rotations from −2.34° to −9.30° in the present study. In particular, the mean settlement angles were smaller in F. albicostatus than those of C. challengeri. This result suggests that the two species tend to avoid wave impact settling on the substrate at vertical settlement angles, particularly F. albicostatus. According to our observations and measurements, F. albicostatus with a relatively tight adhesional capacity due to its large basal plate but an extended height and less streamlined shell base showed a smaller mean angle than C. challengeri with a relatively weak adhesional capacity due to its small basal plate but a short height and more streamlined shell base (Fig. 2). Some studies have suggested that organisms change their body orientation to minimize frontal areas or adopt streamlined shapes, limiting movement during strong flows (Denny, 1985; Weissburg et al., 2003). The present study serves as an important reference for supporting the hydrodynamic shell orientation pattern of barnacles.
Biomimicry is a branch of science that emulates sustainable natural strategies using an interdisciplinary approach involving biologists, physicists, chemists, engineers, architects, and designers (Knippers et al., 2016). This scientific approach is an excellent and creative way to scrutinize sustainable solutions to human problems by mimicking nature in its analogies, phenomena, and patterns (Aziz & El sherif, 2016). For example, nature provides inspiration for innovative materials and structures with optimal morphologies and topologies to produce brilliant mechanical and structural properties and resilient options for industries (Ahamed et al., 2022). Living organisms provide smart ideas for biologists and engineers to develop sustainable technologies. Marine animals (e.g., invertebrates) are frequently used as model species for biomimicry because of their great biodiversity. Natural adhesives have been developed from barnacles. However, their morphometric characteristics against strong wave forces have not yet been utilized to develop innovative technologies in the design, architecture, and motor and airplane industries. Therefore, it is necessary to consider the ideal morphometric characteristics of barnacles as a potential target for biomimicry to avoid strong waves. To date, few biomimicry studies have examined the morphometric characteristics of marine organisms (e.g., fish and whales). Engineers have shown interest in the carapace of boxfish, whose shape exhibits impressive hydrodynamic characteristics to develop new vehicles (Chowdhury et al., 2019). Engineers use design principles from humpback whale flippers, especially their tubercles, to increase the efficiency and power generation of wind turbines (Krishnan et al., 2023). In conclusion, bioinspired hydrodynamic or aerodynamic techniques learned from living marine organisms, such as barnacles, could significantly enhance the efficiency and reliability of a variety of engineering divisions (e.g., design, transportation, architecture, civil engineering, and electric power).
Author Contributions
Conceptualization: BJK. Data curation: JYK, BJK. Formal analysis: JYK. Funding acquisition: BJK. Investigation: JYK, BJK. Methodology: BJK. Project administration: BJK. Resources: JYK, BJK. Software: JYK. Supervision: BJK. Validation: JYK, BJK. Visualization: JYK, BJK. Writing – original draft: BJK. Writing – review & editing: JYK, BJK.
Funding
This study was supported by the National Institute of Ecology and was funded by the Ministry of Environment (MOE) of the Republic of Korea (No. NIE-B-2025-18 and No. NIE-B-2025-44).
Figures and Table
Fig. 1
Photos of barnacle species in this site on Daejang Island. (A) Two different species inhabiting a plot (30×30 cm) together (scale bar=3 cm). (B) Close-up image of Chthamalus challengeri. (C) Close-up image of Fistulobalanus albicostatus.
Fig. 3
The percent cover (%) of Chthamalus challengeri and Fistulobalanus albicostatus in this study (n=5 plots for each species). Boxes: interquartile ranges; horizontal lines: medians; and whiskers: ranges.
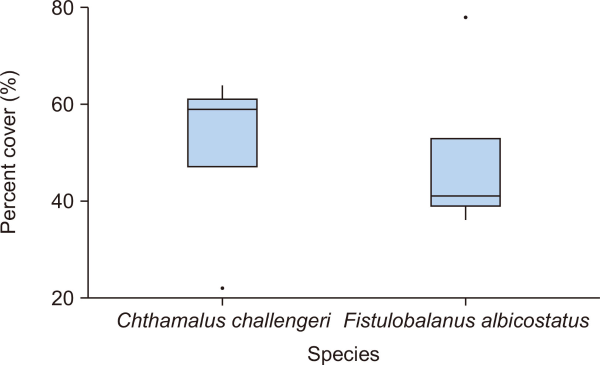
Fig. 4
The density (inds./cm2) of Chthamalus challengeri and Fistulobalanus albicostatus in this study (n=5 subplots for each species). Boxes: interquartile ranges; horizontal lines: medians; whiskers: ranges; and dots: possible outliers.
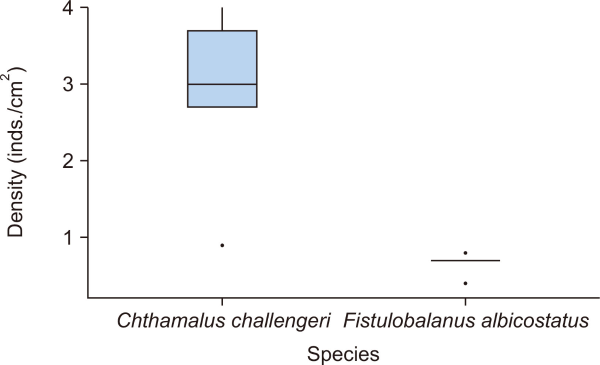
Fig. 5
Shell size of two barnacle species, Chthamalus challengeri and Fistulobalanus albicostatus (n=50 inds.). (A) Shell length (mm); (B) shell width (mm); and (C) shell length-to-width ratio. Box plots display the interquartile range, median (horizontal line), ranges (whiskers); and possible outliers (dots).
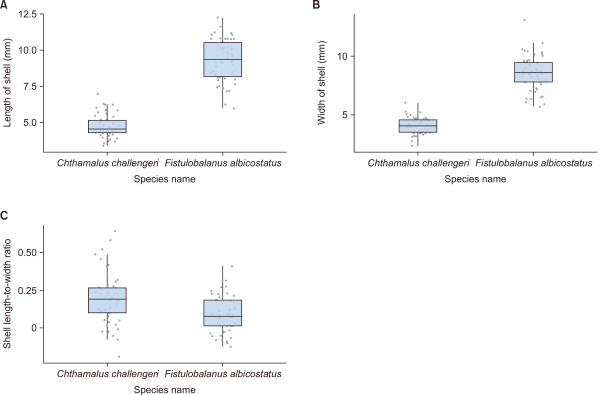
Fig. 7
Frequency plots showing the distribution of settlement orientation angles (°) in Chthamalus challengeri (A) and Fistulobalanus albicostatus (B) (n=50 for each species). The angle represents the vertical direction of shell orientation relative to wave direction. The mean±standard deviation is −9.3±39.8° and −2.3±29.0° for C. challengeri and F. albicostatus, respectively.
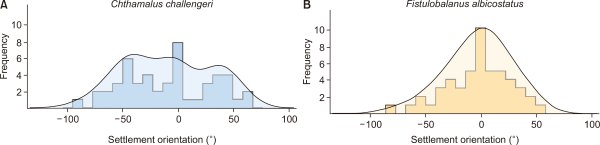
Table 1
Results of one-way analysis of variances comparing percent cover, density, size, and Mann-Whitney U-test comparing settlement orientation between two barnacle species
| Variable | F | df₁ | df₂ | P-value |
|---|---|---|---|---|
| Percent cover | ||||
| Percent cover (%) | 0.01 | 1 | 8.0 | 0.915 (NS) |
| Density | ||||
| Density (inds./cm²) | 16.27 | 1 | 4.08 | <0.05 |
| Size | ||||
| Shell length (mm) | 377.41 | 1 | 75.1 | <0.001 |
| Shell width (mm) | 377.34 | 1 | 71.3 | <0.001 |
| Length-to-width ratio | 12.69 | 1 | 89.1 | <0.001 |
| Variable | Statistic | df | P-value | |
| Settlement orientation | ||||
| Settlement orientation (°) | 1,095 | 0.287 (NS) |