
- P-ISSN 1225-0163
- E-ISSN 2288-8985





Bisphenols and phthalates are endocrine-disrupting chemicals that are commonly used in packaging and as plasticizers. However, they pose health risks through ingestion, inhalation, and dermal contact. Accurate analysis of these pollutants is challenging owing to their low concentration and their presence in complex oil matrices. Therefore, they require efficient extraction and detection methods. In this study, an analytical method for the simultaneous quantification of bisphenols and phthalates in corn oil is developed. The dynamic multiple reaction monitoring mode of liquid chromatography-tandem mass spectrometry is used according to the different polarities of bisphenols and phthalates. The method is validated by assessing system suitability, linearity, accuracy, precision, homogeneity, and stability. The determination coefficients are higher than 0.99, which is acceptable. The percentage recovery and coefficient of variation of the accuracy and precision confirm that this analytical method is capable of simultaneously quantifying bisphenols and phthalates in corn oil. The bisphenols and phthalates in the formulations and pretreatment samples are stable for 7 d at room temperature and 24 h in an auto-sampler. Therefore, this validated analytical method is effective for the simultaneous quantification of bisphenols and phthalates in oils.
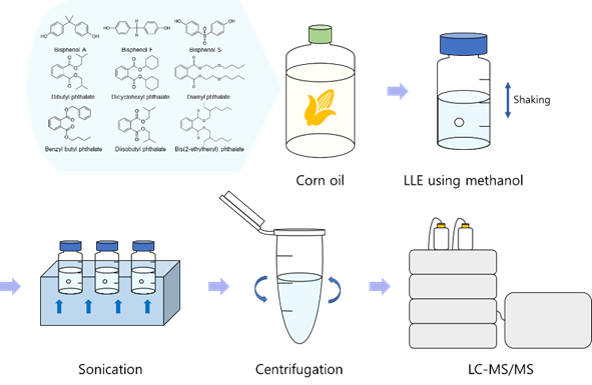
Bisphenols and phthalates are organic pollutants that occur in the environment and consumer products.1 Bisphenols are used in packaging, adhesives, and internal coatings of cans, and phthalates are plasticizers used in the handling and storage of food, such as in appliances and containers for cooking food.2,3 These compounds are endocrine-disrupting chemicals (EDCs), commonly referred to as environmental hormones.3 Humans are exposed to EDCs during eating, breathing, or skin contact.4 In addition, pollutants exposed to the environment can be absorbed by animals through food or air and are potentially transferred to humans through the food chain.2 Some of them are known to have effects such as reproductive problems and endocrine disruptions, and toxicity may accumulate and affect future generations.1,2,5 Because EDCs are mainly soluble in fat, they are not easily decomposed, unlike common natural hormones, and are absorbed and accumulated in fat and tissues of the body.1,6 Analysis in oil is difficult and necessitates analytical techniques with low detection limits owing to the low concentration of analytes. The lipophilic properties of a material and properties of the matrix considerably affect extraction efficiency, recovery, and precision.1 The fat in samples with high oil content must be removed prior to injection into the detection instrument in order to increase the efficiency of the chromatography column. Liquid-liquid extraction (LLE) and solid phase extraction are performed to determine analytes by removing fat.7 According to previous studies, gas chromatography (GC),8 GC-mass spectrometry (GC-MS),1-4,6-11 and liquid chromatography-mass spectrometry (LC-MS)3,5,12 were performed to measure the concentration of bisphenols and phthalates. A derivatization step was included in the method for analyzing bisphenol using a mass spectrometer to increase the sensitivity of detection and enhances the physicochemical properties of bisphenol. N,O-bis (trimethylsilyl) trifluoroacetamide with 1 % trimethylchlorosilane was used as the derivative reagent, and the compound was quantified by GC-MS.2,10
Through GC-MS with this derivatization method, phthalates showed the same (m/z 149) and several non-specific ion values, thereby enabling quantification. However, because bisphenol S (BPS) was not detected, a pretreatment method that included derivatization and simultaneous analysis using GC-MS was not suitable. In particular, phthalate analysis is limited by the use of organic solvents and presence of plastic products and gases, which might exist in the laboratory even when using products without phthalates and glassware during pretreatment. In addition, the processes of extracting, purifying, and concentrating samples are time-consuming and increase the risk of contamination.1 Therefore, a simple pretreatment method must be performed prior to injecting the sample into the instrument to minimize contamination. To this end, here, LLE using methanol was selected as the pretreatment method for the simultaneous measurement of bisphenols and phthalates in corn oil. Furthermore, simultaneous analyses were performed through LC-MS/MS. In this study, three bisphenols (BPA, BPS, BPF) and six phthalates (BBP, DBP, DEHP, DCHP, DIBP, DnPP), as shown in Fig. 1, were selected and spiked into corn oil to evaluate the applicability of the developed analytical method. This selection was driven by the increasing concern over the toxicity of BPA, leading to heightened interest in alternative substances such as BPS and BPF, as well as the inclusion of these compounds in domestic and international regulatory frameworks due to their widespread detection.13,14
The standards for bisphenol A (BPA, 100 %), bisphenol F (BPF, 99.3 %), BPS (99.4 %), benzyl butyl phthalate (BBP, 98.8 %), dibutyl phthalate (DBP, 99.6 %), bis(2-ethylhexyl) phthalate (DEHP, 99.7 %), dicyclohexyl phthalate (DCHP, 99.0 %), diisobutyl phthalate (DIBP, 99.7 %), and dipentyl phthalate (DnPP, 99.5 %) were purchased from Sigma-Aldrich (Saint Louis, MO, USA). BPA (BPA-13C12, 100 μg/mL in acetonitrile) and bis(2-ethylhexyl) phthalate-3,4,5,6-d4 solutions (DEHP-d4, 100 μg/mL in methanol) were purchased from Chemservice (West Chester, PA, USA) and Accustandard (New Haven, CT, USA), respectively, and used as internal standards (ISs). Methanol and water were purchased from Merck (Darmstadt, Germany). Ethyl alcohol and hexane were purchased from Burdick and Jackson (Brooklyn, NY, USA), respectively. Corn oil was purchased from Daejung (Gyeonggi-do, Korea) and used as an excipient to verify the analytical method.
The stock solutions of the three bisphenols and six phthalates were prepared by dissolving the pure standards in methanol and refrigerating them. After preparing a solution with a concentration of 1 − 1000 ng/mL using the standard stock solution and methanol, corn oil was added at a dilution ratio of 10 to prepare a standard solution for the calibration curve. The concentration range of the calibration curve was 5 − 100 ng/mL for BPA and BPF; 0.1 − 100 ng/mL for BPS; 0.1 − 100 ng/mL for DBP, DCHP, and DnPP; 0.5 − 100 ng/mL for BBP; 1 − 100 ng/mL for DIBP; and 10 − 100 ng/mL for DEHP.
BPA-13C12 and DEHP-d4 were used as the ISs of the bisphenols and phthalates, respectively, and prepared at concentrations of 1000 and 500 ng/mL using methanol. Then, they were added to the volume of the calibration curve concentration and diluted 10 times.
At the beginning of the study, the samples treated with corn oil were diluted with hexane and acetone. After concentrating the diluted solution, corn oil remained. As the sample was continuously injected into the MS system and analyzed, the retention time of the analytes decreased, and the efficiency of the analysis column decreased.
Therefore, during the extraction process, it was necessary to use a solvent in which the analyte could be extracted without leaving corn oil. Therefore, ethyl acetate, dichloromethane, chloroform, ethyl ether, acetonitrile, methanol, and ethanol were considered as potential solvents. Because a large amount of corn oil remained in most solvents except methanol, LLE using methanol was selected as the extraction method for the simultaneous measurement of bisphenols and phthalates. To minimize phthalate contamination, 0.1 and 1 mL of corn oil and methanol, respectively, were added to a 2 mL glass vial to perform LLE for 10 min, after which ultrasonic extraction was performed for 15 min. The solution was centrifuged at 8,000 g for 5 min to dilute and mix 0.1 mL of the supernatant, 0.1 mL of the IS, and 0.8 mL of methanol. All glassware was cleaned with hexane and acetone prior to use, followed by drying at approximately 180 °C for a minimum of 4 hours.1,4,5,11,12 The solvent used in this study was verified to have no interference that could affect the quantification of the analytes. Additionally, the blank control, which did not contain the three bisphenols or six phthalates, showed no interference during the sample pretreatment process.
In this study, 1260 Infinity HPLC and 6460 Triple Quad MS systems (Agilent Technologies, Palo Alto, CA, USA) were used to analyze bisphenols and phthalates. Upon using the mobile phase to which the buffer solution was added, bisphenols were not detected. Thus, pure water and methanol were used as the mobile phase for separating bisphenols and phthalates, respectively. A Poroshell 120 EC-C18 column (2.7 µm, 2.1 mm × 100 mm, Agilent Technologies, Palo Alto, USA) was used. The analysis was performed in the electrospray ionization and dynamic multiple reaction monitoring modes. The analysis conditions are listed in Table 1.
Method validation was performed to confirm the applicability of the method in compliance with the ICH Q2(R1) and European SANTE/11813/2017 guidelines.15,16 Quality control (QC) samples completely dissolved in corn oil were prepared for a recovery test to evaluate accuracy and precision. The standard solution was diluted with ethyl alcohol and hexane to ensure complete dissolution in corn oil, followed by treatment with corn oil. The QC concentrations of 1, 2, and 10 μg/mL were used to represent the low, medium, and high QCs (LQC, MQC, and HQC), respectively. Three replicates per concentration were analyzed through a pretreatment process to confirm within-run variations and two or more times on different dates using the same method to confirm between-run variations. Accuracy and precision were evaluated by measuring the recovery and coefficient of variation (CV), respectively. After preparing the formulations of the QC samples with concentrations of 1 and 10 μg/mL, they were stored at room temperature for 7 d, and their stability was confirmed. The initial concentration, which was the standard for the stability test, was confirmed from the results of the homogeneity test by analyzing the concentration in the upper, middle, and lower layers of the test solution. In addition, to confirm the stability after pretreatment, the sample was pretreated, stored in an auto-sampler of the instrument for 24 h, and analyzed. System suitability was evaluated on the basis of the precision of the response value obtained by repeatedly injecting the lowest concentration of the calibration curve six times. Linearity was evaluated using a calibration curve, and the accuracy and precision of the calibration curve were evaluated using the relative error (RE) and CV, respectively. The RE and CV were calculated using the following equations:
RE (%) = [(measured concentration – expected concentration) / expected concentration] × 100
and
CV (%) = [standard deviation of measured concentration/average value of measured concentration] × 100
Most previous studies analyzed bisphenols and phthalates through LC- and GC-MS, respectively, instead of analyzing them in one instrument at the same time.3,17 In several studies, only phthalates and BPA or BPF have been simultaneously analyzed through GC-MS.2,6 In this study, to simultaneously analyze three bisphenols and six phthalates, a GC-MS analysis method using derivatization was first selected. However, the desired result was not obtained and this was confirmed by verifying analysisusing LC-MS/MS. According to previous studies, 10, 2, and 2 mM ammonium acetate in acetonitrile (pH 4), water, and methanol, respectively, were used as the mobile phases to analyze phthalates.5,12 To analyze BPA, pure water and methanol were used as the mobile phases.17-20 In this study, we attempted to separate and analyze bisphenols and phthalates using a mobile phase containing a buffer solution. However, BPF and BPS were not detected even when using various buffer solutions. Therefore, pure water and methanol were selected as the mobile phases, and it was confirmed that all bisphenols were detectable and suitable for the detection of phthalates.
To evaluate the suitability of the system for this analysis method, standard solutions of BPA, BPF, BPS, DBP, DCHP, DnPP, BBP, DIBP, and DEHP at the lowest concentration level of the calibration curve were injected into the LC-MS/MS system six times. It was confirmed that the precision of the response value was 1.1 − 9.1 %, which was within the evaluation standard of 15 % (Table 2). The LC-MS/MS instrument used to verify the analytical method was maintained to analyze the lowest concentration of the calibration curve.
To confirm linearity, calibration curves were constructed by preparing at least five concentrations of samples for BPA, BPF, BPS, DBP, DCHP, DnPP, BBP, DIBP, and DEHP. The weight for the calibration curves was 1/x (Wan et al., 2013), and the determination coefficients (r2) ranged from 0.99368-0.99734 (Fig. 2).
In addition, the accuracy and precision of the calibration curve of each compound were -13.0 % – 9.5 % and 0.5 % – 9.8 %, respectively (Table 3). Each calibration curve showed a correlation coefficient of ≥0.99 and result within 15 % of the appropriate criteria for accuracy and precision. Therefore, this method was determined to be suitable for quantifying all compounds.
Accuracy and precision were measured in three repetitions for concentrations of 1 (LQC), 2 (MQC), and 10 (HQC) μg/mL over 3 d; within- and between-run variations were evaluated in terms of recovery and CV (Table 4). The appropriate criteria for accuracy and precision were a recovery within 100 ± 20% and CV within 20 %, respectively. The results of the BPA, BPF, BPS, DBP, DCHP, DnPP, BBP, DIBP, and DEHP analyses all met these criteria. Therefore, it was determined that this method was appropriate.
To evaluate the stability of the formulation, the homogeneity was evaluated on the day of preparation to measure the initial concentration, which was used as the basis for content change. The homogeneity was measured by sampling the upper, middle, and lower layers of the 1, 10 μg/mL formulations, respectively, once; the results are shown in Table 5. The homogeneity of the formulation containing bisphenols and phthalates was determined to be well prepared within 100 % ± 20 %, with a CV of 20 %, which was a suitable criterion with a content of 87.1 %–108.8 % and CV of 0.5 % – 7.5 %. In addition, the analysis was performed after 24 h of storage in the instrument after pretreatment to confirm the stability before analysis after the sample was prepared. To confirm whether the formulation was stable at room temperature, its stability was evaluated by comparing the initial and analyzed concentrations after storage at room temperature for 7 d (Table 6). The stability after pretreatment and 7 d at room temperature was 88.3 % – 115.9 % and 95.6 % – 117.8 %, respectively, which were within 20 % of the initial concentration. These results indicate that suitable criteria for stability were met and that the formulation was stable for 24 h and 7 d at room temperature.
The concentrations of various bisphenols and phthalates in corn oil were measured using an analytical method that involved evaluating the system suitability, linearity, accuracy, homogeneity, and stability of the tested compounds via LC-MS/MS. In this method, LC-MS/MS was appropriately maintained to analyze the minimum concentration level of the calibration curve. The r2 value of the calibration curve was at least ≥0.99, and the accuracy and precision of the calibration curve were good. The accuracy and precision of the QC samples evaluated in terms of recovery and CV were high, and the formulation was stable before injection into the instrument after pretreatment and exposure to room temperature. The proposed method can be applied for the simultaneous quantification of BPA, BPF, BPS, DBP, DCHP, DnPP, BBP, DIBP, and DEHP in corn oil and can be used in related applications for measuring bisphenols and phthalates in oil.
J. Pereira, M. C. Selbourne, and F. Pocas, Food Control., 98, 54-60 (2019). https://doi.org/10.1016/j.foodcont.2018.11.003 https://doi.org/10.1016/j.foodcont.2018.11.003
O. Ballesteros, A. Zafra, A. Navalon, and J. L. Vilchez, J. Chromatogr. A, 1121(2), 154-62 (2006). https://doi:10.1016/j.chroma.2006.04.014 https://doi.org/10.1016/j.chroma.2006.04.014
H. T. Wan, P. Y. Leung, Y. G. Zhao, X. Wei, M. H. Wong, and Chris K. C. Wong, J. Hazard. Mater., 261, 763-769 (2013). http://dx.doi.org/10.1016/j.jhazmat.2013.01.034 https://doi.org/10.1016/j.jhazmat.2013.01.034
R. Babu-Rajendran, G. Preethi, R. K. Poopal, N. P. Nikhil, K. Vimalkumar, A. Subramanian, and S. Krishna-Kumar, J. Chromatogr. B, 1079, 15-24 (2018). https://doi.org/10.1016/j.jchromb.2018.01.039 https://doi.org/10.1016/j.jchromb.2018.01.039
P. Vinas, N. Campillo, M. Pastor-Belda, A. Oller, and M. Hernandez-Cordoba, J. Chromatogr. A, 1376, 18-25 (2015). http://dx.doi.org/10.1016/j.chroma.2014.12.012 https://doi.org/10.1016/j.chroma.2014.12.012
S. Lopez-Feria, R. Lucena, S. Cardenas, and M. Valcarcel, Anal. Bioanal. Chem., 395(3), 737-746 (2009). https://doi:10.1007/s00216-009-3066-4 https://doi.org/10.1007/s00216-009-3066-4
P. Gimeno, S. Thomas, C. Bousquet, A-F. Maggio, C. Civade, C. Brenier, and P-A. Bonnet, J. Chromatogr. B, 949-950, 99-108 (2014). http://dx.doi.org/10.1016/j.jchromb.2013.12.037 https://doi.org/10.1016/j.jchromb.2013.12.037
H. Jung, Y. Hong, D. Lee, K. Pang, and Y. Kim, Environ. Toxicol. Pharmacol., 35(2), 278-283 (2013). http://dx.doi.org/10.1016/j.etap.2013.01.002 https://doi.org/10.1016/j.etap.2013.01.002
H-Y. Shen, Talanta, 66(3), 734-739 (2005). http://doi:10.1016/j.talanta.2004.12.021 https://doi.org/10.1016/j.talanta.2004.12.021
A. Vavrous, J. Pavlouskova, V. Sevcik, K. Vrbik, and R. Cabala, J. Chromatogr. A, 1456, 196-204 (2016). http://dx.doi.org/10.1016/j.chroma.2016.06.014 https://doi.org/10.1016/j.chroma.2016.06.014
M. Hyun, L. Rathor, H-J. Kim, T. McElroy, K. H. Hwang, S. Wohlgemuth, S. Curry, R. Xiao, C. Leeuwenburgh, J-D. Heo, and S. M. Han, Toxicology, 461, 152924 (2021). https:// doi.org/10.1016/j.tox.2021.152924 https://doi.org/10.1016/j.tox.2021.152924
I. Lee, J. Ra, and K. Ji, J. Environ. Health Sci., 47(1):1-19 (2021). https://doi.org/10.5668/JEHS. 2021.47.1.1 https://doi.org/10.5668/JEHS.2021.47.1.1
B-C. Lee, H. Yoon, B. Lee, P. Kim, H-B. Moon, and Y. Kim, J. Ind. Eng. Chem., 99, 68-73 (2021). https://doi.org/10.1016/j.jiec.2021.03.051 https://doi.org/10.1016/j.jiec.2021.03.051
N. Caballero-Casero and S. Rubio, Anal. Chim. Acta, 1144, 14-25 (2021). https://doi.org/10.1016/j.aca.2020.11.057 https://doi.org/10.1016/j.aca.2020.11.057
N. Dreolin, M. Aznar, S. Moret, and C. Nerin, Food Chem., 274, 246-253 (2019). https://doi.org/ 10.1016/j.foodchem.2018.08.109 https://doi.org/10.1016/j.foodchem.2018.08.109
L. K. Sosvorova, T. Chlupacova, J. Vitku, M. Vlk, J. Heracek, L. Starka, D. Saman, M. Simkova, and R. Hampl, Talanta, 174, 21-28 (2017). http://dx.doi.org/10.1016/j.talanta.2017. 05.070 https://doi.org/10.1016/j.talanta.2017.05.070