
- P-ISSN 1225-0163
- E-ISSN 2288-8985


To prevent the recidivism of criminals with mental disorders, e.g., bipolar disorder, the Republic of Korea introduced the therapeutic order system in 2016 to ensure that these individuals receive systematic treatment and management, including medication administration and regular monitoring of treatment compliance. However, the International Organization for Standardization (ISO) requires laboratories performing quantitative analysis to include measurement uncertainties in the quantitative results to ensure reliable outcomes. Therefore, in this study, the measurement uncertainty of the determination of Li in urine using ion chromatography (IC) was established. The standard addition method of weighted calibration without an internal standard was employed to quantify urinary Li. Sample preparation involved centrifuging the urine and diluting the supernatant with deionized H2O, and the Li+ in the urine was analyzed using IC. The potential factors that contribute to the uncertainty of the final result were examined and evaluated. The major contributing factors influencing the combined standard uncertainty were the calibration curve and repeatability, whereas the preparation of the standard solution and sample dilution were minor factors. The relative expanded uncertainty of the measured concentration in a forensic urine sample was 35.3 %.
Since the 1970s, mood stabilizers, such as lithium (Li), valproate, and carbamazepine, have been widely used as primary medications in treating bipolar disorder. Li, which is administered as a carbonate salt, is effective in treating acute mania and suppresses its recurrence via long-term treatment.1 Continuously monitoring the intake of Li is crucial in preventing the worsening of mental disorders and potential harm to others that may occur if the medication is discontinued arbitrarily.2 To prevent the recidivism of criminals with mental disorders, e.g., bipolar disorder, schizophrenia, depression, anxiety, and alcohol use disorders, and protect the public interest, Ministry of Justice Korea Correction Service introduced the therapeutic order system in 2016 to ensure that these individuals receive systematic treatment and management, including medication administration and regular monitoring of treatment compliance. This is a problem-solving system that focuses on rehabilitation and preventing recidivism, emphasizing treatment instead of the traditional criminal justice approach to reduce the number of violent crimes committed by persons with mental disorders.3 Medication screening is conducted periodically to ensure that a probationer with a mental illness is receiving appropriate treatment and care, and the biological sample commonly analyzed in such a case is urine. Urine is useful in drug analysis because it is easier to collect than blood and relatively large amounts of parent drugs and their metabolites are excreted in the urine. To measure the Li+ concentrations in such biological samples, analytical methods, such as spectrophotometry,1 electrothermal atomic absorption and inductively coupled argon plasma atomic emission spectrometry,4-6 and ion chromatography (IC),7,8 are utilized.
The International Organization for Standardization (ISO, Geneva, Switzerland) requires laboratories performing quantitative analysis to adhere to the international standard ISO/IEC 17025, which mandates the inclusion of measurement uncertainties in the quantitative results to ensure confidence and reliability. This process confirms the potential sources of error during the experiment and enables the calculation of the measurement uncertainty caused by each factor. This enhances the reliability of the analytical results by clearly identifying the main factors contributing to errors and enables the reduction of errors associated with each factor through improvement processes.9,10 In this study, quantitative results of Li+ in the urine samples of probationers treated using Li2CO3 for bipolar disorder were performed using IC, and potential factors affecting the measurement results were examined. Calibration curve was constructed using the standard addition method with the weighting factor of 1/x in order to compensate for matrix effects that can interfere with the measurement of the analyte. The urine was prepared using the dilute-and-shoot approach, followed by separation and quantification to measure the Li+ concentration. The measurement uncertainties of the obtained results were then calculated and evaluated.
Li+ standard solution (0.997 ± 0.005 mg/mL in HNO3, TraceCERT) and methanesulfonic acid (MSA, 0.4 ± 0.008 M) used in producing the mobile phase were respectively purchased from Sigma-Aldrich (St. Louis, MO, USA) and Thermo Fisher Scientific (Waltham, MA, USA). All solutions were prepared using deionized H2O obtained using a Direct-16 MilliQ water purification system (MilliporeSigma, Burlington, MA, USA). The purchased standard solution was stored at 24 ± 4 ℃ until use.
The sample used in Li+ measurement was the urine of a probationer with bipolar disorder, and its evaluation was requested by a metropolitan area probation office. The urine was stored at 4 ℃ until analysis.
QC samples were prepared to yield Li+ concentrations in urine of 2, 6, 60, and 300 ng/mL. These samples, which were prepared in 5 aliquots of each concentration, were measured repeatedly over 3 d to determine the precision of the analytical method.
A calibration curve was constructed using the standard addition method without an internal standard, with the calibrators prepared at concentrations of 2, 10, 50, 100, 200, and 400 ng/mL. The weighting factor (wi) applied in the construction of the calibration curve was 1/x. The area of the Li+ peak in the obtained chromatogram was determined, and this value was then used as the y-axis of the calibration curve.
Urine (100 μL) that was centrifuged at 5500 g for 5 min was transferred to a 1.5 mL polypropylene tube. Subsequently, 900 μL of deionized H2O was added to the sample, which was mixed, and then 25 µL of the mixture was injected into the IC instrument for analysis.
A Dionex Integrion HPIC system (Thermo Fisher Scientific) equipped with a conductivity detector, reagent-free IC-eluent regeneration controller, column heater, and Dionex AS-AP autosampler with a 25 μL sample loop was utilized in Li+ quantification. The program used in data processing was Chromeleon 7.2 SR5 (Thermo Fisher Scientific). Guard (Dionex IonPac CS12A-8 μm Guard, 4 × 50 mm, Thermo Fisher Scientific) and analytical columns (Dionex IonPac CS12A-8 μm, 4 × 250 mm, Thermo Fisher Scientific) were utilized to effectively separate and analyze Li+ from mono- and divalent cations and interfering substances within the urine matrix, and the column temperature was maintained at 35 °C. MSA (20 mmol/L) was used as the mobile phase under isocratic condition and the flow rate was set at 1.0 mL/min. The sample injection volume and run time required were 25 µL and 15 min, respectively.
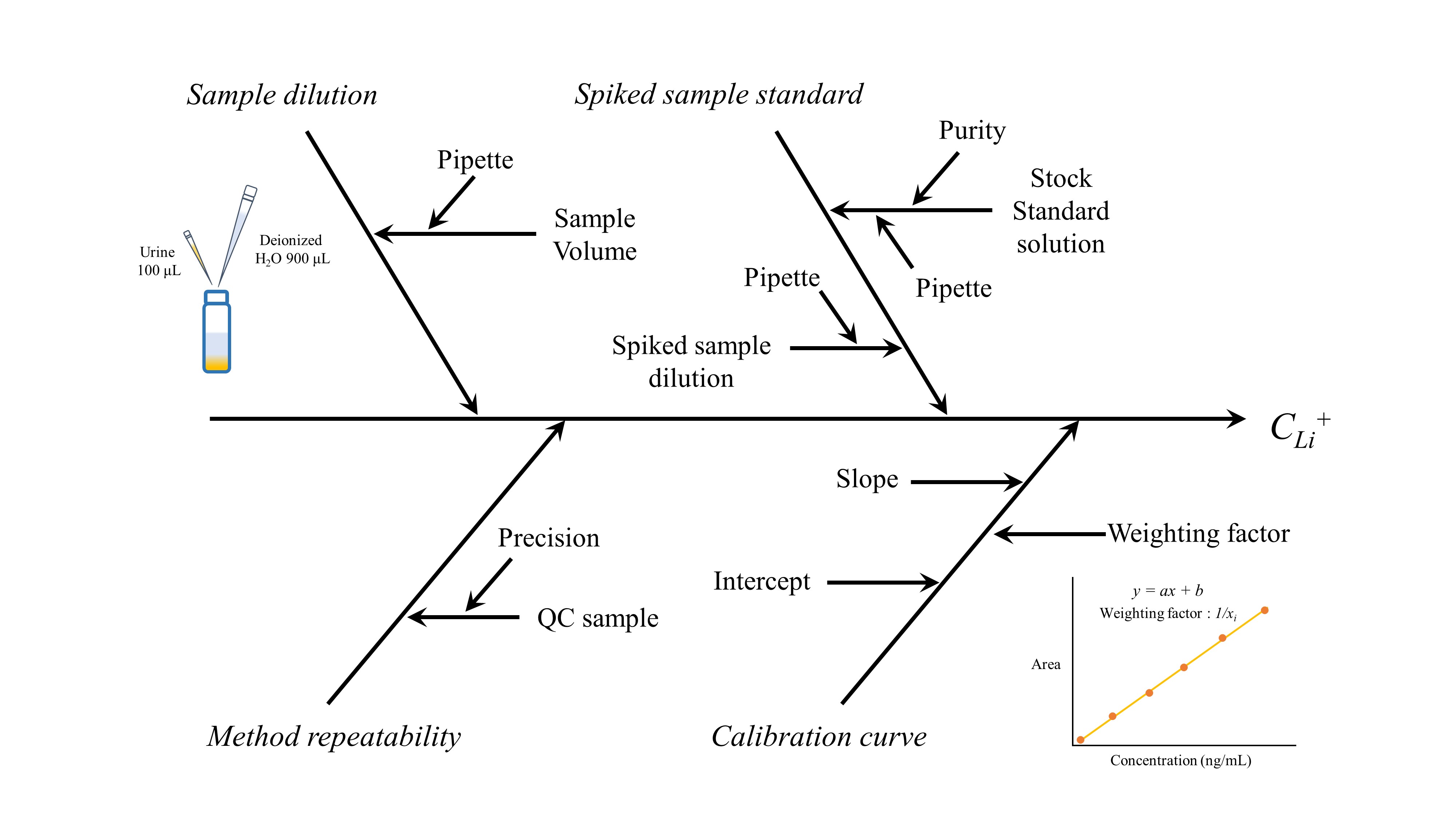
The factors associated with the measurement uncertainty of the Li+ concentration in urine include the uncertainties arising from sample dilution, calibration curve construction, the preparation of the working standard solution, and repeated measurements. These four factors and their characteristic diagram are shown in Fig. 1.
The measurand is defined as the Li+ concentration (ng/mL) in each urine sample, and it is expressed by the following equation :
Cx = CS + δD + δS + δC +δR
where Cx, CS, δD, δS, δCand δR are the Li+ concentration in the urine, the Li+ concentration in the given sample volume, and additive factors for the uncertainties related to sample dilution, spiked sample standard preparation, the calibration curve, and method repeatability, respectively.
To calculate and evaluate the measurement uncertainty, the standard uncertainty was derived from individual uncertainty factors, and the law of uncertainty propagation was applied to the measurement model to yield the combined standard uncertainty. The expanded uncertainty was calculated by multiplying by the coverage factor (k) equivalent to a confidence level of 95 %. The degrees of freedom of the standard deviation by factor were determined using the WelchSatterthwaite equation to calculate the effective degrees of freedom (Veff)
The uncertainty that occurs during the aliquoting and 10-fold dilution of urine (100 µL) is caused by factors related to the pipette. The uncertainty of the pipette during volume measurement was derived from the uncertainty of the calibration certificate. The uncertainties (at a confidence level of approximately 95 %, k = 2) indicated on the calibration certificates of the pipettes (100 and 1000 µL) were (100 ± 0.25) and (1000 ± 2.5) µL, respectively. The standard uncertainty of pipettes (100 and 1000 µL) are 0.125 and 1.25 µL respectively. The relative standard uncertainty (ur(VP1000)) when measuring 900 µL using a pipette (1000 µL) was 0.001388889. The relative standard uncertainty (ur(D)) during sample dilution and effective degree of freedom were 0.00186856 and ∞, respectively
The uncertainty indicated on the Li+ standard solution certificate (20 °C, k = 2) was (0.997 ± 0.005) mg/mL. The standard uncertainty (u(RLi)) of the Li+ standard solution and the relative standard uncertainty (ur(RLi)) were 0.0025 mg/mL and 0.00250752, respectively.
Spiked sample standard of Li+ was finally prepared by spiking working standard solution to pooled blank urine samples at a concentration level of 1 μg/mL. This standard was prepared by 3-fold serial dilution of the stock standard using blank urine with two different pipette of 100 (n = 4) and 1000 μL (n = 4). The relative standard uncertainty (ur(S)) of the preparation process and effective degree of freedom were 0.0025076 and ∞, respectively.
The calibration curves were prepared by plotting the peak area of the target compound against the concentration, which required the preparation of calibrators (n = 6). These calibration curves were used to estimate the target compound concentrations in the urine samples. The calibration functions were calculated and fitted using a linear regression model with a weighting factor (wi), yielding the following equations:
yj = aw + bw · xj
yj = A
where xi and yi are the concentration and peak area of the target compound (A) in the urine sample, respectively.
A urine sample of the Li user was analyzed, yielding in xj = 86.9 ng/mL. The standard uncertainty and relative standard uncertainty of Li+ were obtained by solving regression equations for the weighted regression model. The standard and relative standard uncertainties of Li+ were determined by solving the regression equations of the weighted regression model. The standard uncertainty (u(C)) of the Li+ calibration curve was 8.26033, and the relative standard uncertainty (ur(C)) was 0.0950556, which is calculated as follows11:
where yj is the instrumental response to the analyte, xj is the determined concentration of the analyte, and wj is the weighting factor of the calibration curve for a urine sample obtained from a probationer.
To confirm the repeatability of the measurement results, the uncertainty was calculated from repeated measurements to assess the precision over 3 d, using QC sample aliquots (n = 5) prepared at concentrations of 2, 6, 60, and 300 ng/mL.
The measured averages and standard uncertainties obtained using the QC samples with different concentrations are shown in Table 1. The relative standard uncertainty (ur(R)) of the reproducibility of the measurement results and the degree of freedom were 0.1488924 and 39, respectively.
si, ni, and are the standard deviation of the result of aliquot measurement, number of aliquots, and average of the ni aliquot measurement results, respectively.
The relative standard uncertainties of the uncertainty factors, which were used to calculate the expanded uncertainty, are summarized in Table 2. The effective degrees of freedom were determined using the uncertainties of the individual factors. The relative combined standard uncertainty (urc(C)) of the analyzed substance of 0.1766757 was calculated using the relative standard uncertainties of the individual components. At a confidence level of 95 % with a coverage factor (k) of 2.08, the calculated relative expanded uncertainty (Ur(C)) is 0.3672476.
Ur(C) = urc(C) ×k
Consequently, the Li+ concentration in the urine of an individual using Li2CO3 (a treatment for bipolar disorder) was measured at a 95 % confidence level, with an uncertainty of (86.9 ± 30.7) ng/mL at k = 2. The uncertainty of the measurement of the Li+ concentration is 35.3 %.
This study calculated the measurement uncertainty that arises when measuring the Li+ concentration in urine using IC. The measurement uncertainty of Li+ quantified via the urinalysis was 35.3 %, expressed as a percentage of the measured value. Assessing the sources of uncertainty confirmed that among the four factors contributing to precision – sample dilution, spiked sample standard preparation, the calibration curve, and method repeatability – the levels of precision of method repeatability and the construction of the calibration curve exhibited the largest effects on the uncertainty. The error due to the precision of the method repeatability was determined by calculating the relative standard uncertainty using the measured values of the QC samples at four concentrations (2, 6, 60, and 300 ng/mL). The calculated deviations of 0.03449, 0.08244, 0.09134, and 0.07642 indicated that the uncertainties did not differ significantly. Therefore, all four concentrations used in the QC measurements were included in the uncertainty, and this result was identified as the largest source of uncertainty. The second-largest source of uncertainty was the error arising from the calibration curve, with a relative standard uncertainty of 0.0950556. The absence of internal standards, such as isotope-labeled internal standards, could not minimize the influence of the matrix effect, ultimately leading to an increase in error during quantitative analysis. Due to the unavailability of the internal standard method when measuring the Li+ concentration in urine, the standard addition method was employed. To improve analytical methods in the future, reducing errors by identifying candidate substances that can be used as internal standards and applying them to the analysis may be possible. Additionally, the uncertainties arising from the two main factors may be reduced by 1) using a quantitative concentration range centered around the concentration at which the target compound is most distributed in the actual sample and 2) increasing the number of calibrators used in constructing the calibration curve. However, due to the nature of drug analysis, numerous samples must be analyzed rapidly, considering the economic and temporal aspects of the analytical process. Based on the findings of this study regarding the individual factors affecting measurement uncertainty, we shall continue exploring the improvement of analytical methods to enhance the reliabilities of analytical results and minimize the factors contributing to measurement uncertainty.
M. Sheikh, M. Qassem, I. F. Triantis, and P. A. Kyriacou, Sensors, 22(3), 736 (2022). https://doi.org/10.3390/s22030736 https://doi.org/10.3390/s22030736
G. S. Malhi, M. Tanious and S. Gershon, Bipolar Disord., 13(3), 219-226 (2011). https://doi.org/10.1111/j.1399-5618.2011.00918.x https://doi.org/10.1111/j.1399-5618.2011.00918.x
W. Han and S.-Y. Lee, Korean J. Correct. Discourse, 14(3), 241-275 (2020). https://www.dbpia.co.kr/journal/articleDetail?nodeId=NODE10726863 https://doi.org/10726863
B. Sampson, J. Anal. At. Spectrom., 6(2), 115-118 (1991). https://doi.org/10.1039/JA991060011 https://doi.org/10.1039/JA991060011
P. Leflon, R. Plaquet, F. Rose, G. Hennon, and N. Ledeme, Anal. Chim. Acta, 327(3), 301-306 (1996). https://doi.org/10.1016/0003-2670(96)00107-9 https://doi.org/10.1016/0003-2670(96)00107-9
K. Iguchi, K. Usuda, K. Kono, T. Dote, H. Nishiura, M. Shimahara, and Y. Tanaka, J. Anal. Toxicol., 23(1), 17-23 (1999). https://doi.org/10.1093/jat/23.1.17 https://doi.org/10.1093/jat/23.1.17
A. D. Chapp, S. Schum, J. E. Behnke, T. Hahka, M. J. Huber, E. Jiang, R. A. Larson, Z. Shan, and Q.-H. Chen, Physiol. Rep., 6(7), e13666 (2018). https://doi.org/10.14814/phy2.13666 https://doi.org/10.14814/phy2.13666
J.-Y. Kim, W.-Y. Kwon, S.-I. Suh, and M.-K. In, Anal. Sci. Technol., 25(1), 83-90 (2012). https://doi.org/10.5806/AST.2012.25.1.083 https://doi.org/10.5806/AST.2012.25.1.083
W. T. Jeong, S. W. Park, H. H. Noh, H. B. Lim, K. S. Kyung, and S.-H. Ryu, Korean J. Pesticide Sci., 25(2), 82-98 (2021). https://doi.org/10.7585/kjps.2021.25.2.82 https://doi.org/10.7585/kjps.2021.25.2.82
B. Brasil, R.J.N.B. da Silva, M.F. G.F. C. Camoes, andP.A. S. Salgueiro, Anal. Chim. Acta, 804, 287-295 (2013). https://doi.org/l0.1016/j.aca.2013.10.032 https://doi.org/1016/j.aca.2013.10.032