
- P-ISSN 1225-0163
- E-ISSN 2288-8985

Nails are considered suitable biological materials for diagnosing diseases and biomonitoring for human exposure to metals by measuring trace elements levels. Human fingernail samples were collected from volunteers from the Gezira region, Sudan, samples were measured and studied for their elemental content using the X-ray fluorescence (XRF) method, and the data was analysed by PyMca software and SPSS statistical analysis. The data analysis revealed very high concentrations of toxic elements such as Zn, Ni, Pb, Cd, Sr, As, and Se. A comparison was made with published data from different populations. The goal of the current investigation was to evaluate the levels and patterns of trace and toxic elements in the fingernails. Differences and variations in concentrations for the samples were investigated and discussed.
One of the most crucial and necessary conditions for the body's normal functioning is the stability of its chemical composition, as variations in the concentrations of chemical elements brought on by diseases or environmental, occupational, climatic, and geographic factors can result in a wide range of health disorders.1-3 Elements concentration in nail is capable of indicating side effects due to long time exposure to certain type of elements.
Nail consists of layers of keratin tissue that grows from the matrix, a part of nail hidden beneath the cuticle and is produced by living skin cells. In various studies, nail’s elemental composition is affected by gender, age, geographical location and lifestyle.4 It is undeniable that toxic metals are both an environmental hazard and a workplace hazard. The majority of the changes they could bring about in a human body are irreversible and can lead to a number of illnesses, including harm to DNA.5-8 The majority of extremely complex structured materials6 call for effective analytical methods that can provide details on the components of the materials that are dispersed spatially and enable structural analysis.1,9,10 Human nails' elemental concentrations might offer a means of determining a person's nutritional state. The identification of the elemental compositions of young adults' nails may serve as an indication for evaluating their nutritional status because the various elements found in human nails have distinct metabolic functions.11-13
The most often utilized biomarkers for researching human metal exposure include blood, urine, hair, and nails. Numerous research works have proven the effectiveness of toenails as a favored biomarker. Because toenails are non-invasive to collect and offer a comprehensive, long-term record of exposure (2 – 12 months), volunteers are more likely to consent In addition, nails can be more easily identified than bodily fluids or tissues because they frequently contain larger quantities of metal.14 Toenails are less prone to exogenous contamination like bleaching,15 The elemental exposure evaluation may be impacted by dyeing and other cosmetic exposures.16 The elemental and chemical examination of a variety of materials, such as glass, metals, and ceramics, can be done using X-ray fluorescence (XRF).13,17,18 Many elemental analysis methods, including ion chromatography (IC), laser induced breakdown spectroscopy (LIBS), inductively coupled plasma mass spectroscopy (ICPMS), and others, are frequently employed for the study of materials, especially biological samples like teeth, bones, nails, and stones.19,15,20
The proteins that make up human nails are rich in keratin and contain a range of trace elements in proportion to their intake.21,22 Nails can reflect exposure over a considerable amount of time because of their slow growth rate. When compared to other biological samples, nail clippings are simple to gather and preserve.17,23 The goal of this work was to evaluate the usability of XRF, for reliably measuring multiple elements from human nails.
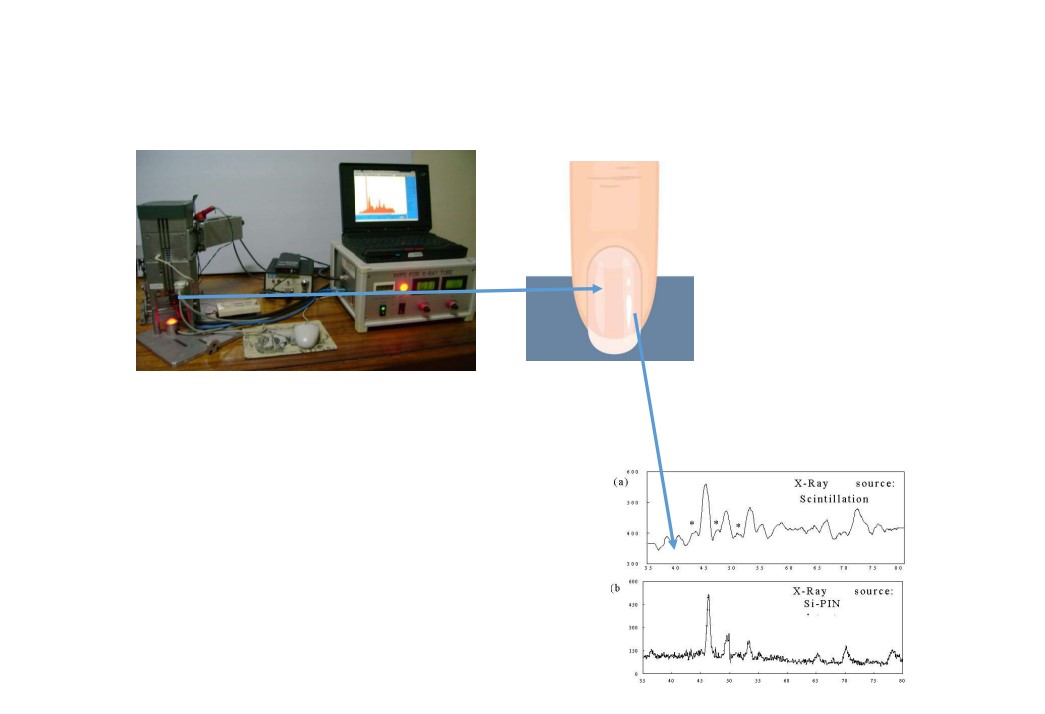
Gathering nail samples from people may appear to be a very easy operation, and there aren't any noticeable temporal or spatial variances. Fingernails samples were collected from volunteers from Gezira region, centre of Sudan (Gezira Scheme24,25), a group of 15 farmers aged between 70 to 18 years in rural areas in Gezira scheme, and since they all frequently utilized fertilizers and pesticides in their farming operations.
Samples were cut with a titanium-coated scissors from the fingernails. The samples were cleaned with copious amounts of water, to remove an inorganic contaminants and then with acetone, to remove any organic contaminants. The samples were then allowed to air dry in a dust-free environment, and placed in individual, spotless plastic containers to prevent any further potential contamination. The nail clippings were then cut in small fragments using titanium coated scissors, to minimise the curvature of the clippings and put in small vials. The fingernails were curved both horizontally and vertically. This the incident beam will not evenly be spread over the nail surface. In addition, bending the nails would compromise the matrix integrity. To avoid these, the nails were cut with a titanium-coated scissors to yield cuttings of 3 mm. Each cutting the nail was analyzed and the concentrations were aggregated.
The development of XRF spectrometry has made it possible to identify elemental elements at tiny scales by combining EDXRF devices with scanning electron microscopy (SEM-EDS). Significantly, XRF is employed with synchrotron radiation sources (SRXRF), which bears a striking resemblance to μ-XRF and has a wide range of applications.19 All measurements and analyses were performed at the multidisciplinary laboratory (MLAB), at ICTP, Italy, A set of portable/ transportable X-ray analytical instruments, Amptek mini X-ray tube (Ag anode, maximum 40 kV, maximum 200 μA) (air cooled with fans) and Amptek Si-Drift detector (energy resolution: 125 eV at 5.9 keV).26
Portable XRF spectrometers are commonly utilized for the chemical characterization of materials. A main beam filter is placed between the sample and the X-ray tube, which serves as an X-ray absorber. Combining kV with an absorption filter produces an energy zone with the ideal Peak/Background ratio. The tube's excitation is adjusted to produce a high integrated intensity above the energy of the element lines of interest's absorption edges on the high-energy side of this region.27,28 To increase the sensitivity of elemental detection, the energy spectra that were available from each clipping were combined prior to analysis. The European Synchrotron Radiation Facility produced the PyMca program, which was used to do the spectral analysis.21,29
The impact of environmental pollution on human health can be investigated through biomonitoring of metabolically inert tissues, like nails, because sample collection, transportation, storage, and analysis preparation are all easily accomplished in this research.30 While geographic location, nutritional state, and environmental factors all affect the concentration of elements in nails, the results were compared with some published values for a sample of healthy adults.2 The majority of the proteins in human nails are keratin rich, and these proteins, through a variety of processes including protein synthesis and chemical interaction with sulfhydryl groups, incorporate trace elements in response to food intakes and other exposures.
Depending on how much is taken in by the body, this protein contains a range of trace elements. Finding other biomarkers that indicate a longer duration of exposure to toxins is desirable because measuring the levels of toxins in blood or urine only represents short-term exposure. Nails can absorb substances over a comparatively longer period of time than can blood or urine samples because of their extremely sluggish growth.19
Fig. 1 is logarithmic scale shows the results of the XRF analysis of the farmers collected samples and as seen from the figure the results displayed that the obtained value in this study revealed that the fingernail light metals levels were found to be in order of S > Cl > K > Ca, however the heavy metals are Pb > Cd > Sr > As > Se. The high accumulation of Pb could have resulted from the exposure to Pb-contaminated food and drinking water as well it seen that Cd, Pb, As and Se are higher than the readings of the other this means that toxicity is potentially very high between the farmers.
The concentration results of the present study of the nails samples which was measured by XRF and calculated by IBM SPSS version 26 software package, is presented in Table 1. In that table “ND” denotes that the element X-ray fluorescence in the nail matrix is not detectable. Of concern the high concentrations of As in the fingernails, especially those of farmers N1 and N2. Such high concentrations are disturbing and alludes to poor health conditions. It should be noted that no inquiries were made as to the then health status of any of the farmers, as these were regarded as private. High concentrations of Se, Sr and Pb were also found in the fingernails of farmer N2. Thus, the farmer’s health status was then, of grave concern. In addition, in the fingernails of farmers N3 the Se, Cd and Pb, the concentrations are very high, and so the Pb concentrations of farmers N7, N8 and N9. On first notice, these high concentrations would have been of grave importance to the general health of the farmers. However, the low concentrations and the absence of these elements in the nail matrices of the other farmers allays fears of a general health hazard.
Table 2 describes the relationships between the variables under study, with the correlation coefficients quantified and the significance of the correlations reported at either the 0.05 or 0.01 level. A significance level of 0.05 (indicated by a single asterisk *) denotes that the correlation is significant at the 0.05 level, meaning there is less than a 5 % probability that the observed correlation is due to chance. In contrast, a significance level of 0.01 (indicated by a double asterisk) indicates that the correlation is significant at the 0.01 level, reflecting less than a 1 % probability that the result is due to chance. Correlations marked with (**) can be described as highly significant, as they demonstrate stronger confidence in the observed relationship between the variables.
The interrelationships between the elements detected in nails are presented in Table 2 some interactions between the elements in nails are presented. In Table 3 the first five columns on the left present the mean, median, standard deviation (STD), minimum and maximum values of the elemental concentrations, however the other nine columns after present the readings of other researchers show the elements' specified concentrations in human nails. They are distinct from one another in all of the research papers that are stated; this may be because sample donors lived in various places with varying trace element exposures.1 Most concentration values determined in this studies compare favorably with those found in previous studies. However, the concentration values of the elements As, Se, Sr, Cd and Pb are orders of magnitude greater than those found in the previous studies. These marked differences are of grave concern. These elements normally occur in ores, which could have been included in the agriceuticals. It can be surmised that these chemicals used, and perhaps still being used, by the farmers are of inferior quality. More so, these high, and thus toxic, concentrations in the nail matrices indicates a poor health conditions. It is recommended that these results be communicated to the Sudanese department of agriculture and that the farmers be taken for medical examination.25
This work represents an encompassing determination of the elemental concentration of the nail matrix. It has been down that agricultural chemicals, environmental and nutritional factors have been demonstrated to have a major impact on the amounts of heavy metals in nail tissue. However, the present paper indicates that high levels of Pb, Cd, As, Ni, Zn and other potential toxic elements found in nail samples from farmers in this area means the need for raising public awareness of the risks that accompany this line of work and enabling these volunteers to take the appropriate safety precautions are urgently needed. Therefore, use of fingernails as biomarkers of exposure to toxic metals supposed to be increased over time by using different experimental techniques. The relatively high concentrations of the inorganic elements found in the nail matrices are indicative of agriceuticals used by the farmers, were of a low quality. It is imperative these findings should be brought to the attention of Ministries of Agriculture in the Sudanese government (sections). More so, these farmers should undergo agriculture instructions/teaching, in which they should be made aware of these health hazards.
S. S. Savinov, R. M. Sharypova, and A. I. Drobyshev, J. Anal. Chem., 75(3), 409-415 (2020). https://doi.org/10.1134/S1061934820030168 https://doi.org/10.1134/S1061934820030168
T. P. Sanches, F. A. Genezini, and M. Saiki, Braz. J. Radiat. Sci., 8(3), 1-12 (2020). https://doi.org/10.15392/bjrs.v8i3.1281 https://doi.org/10.15392/bjrs.v8i3.1281
S. M. Salwa M. Ibrahim, A. M. Abdelbagi, and Al. E. S. ALi E. Sharf Eldeen, J. Anal. Sci. Methods Instrum., 4(1), 1-8 (2014). https://doi.org/10.4236/jasmi.2014.41001 https://doi.org/10.4236/jasmi.2014.41001
E. Koseoglu, B. Kutuk, O. U. Nalbantoglu, R. Koseoglu, and M. Kendirci, J. Trace Elements Med. Biol., 50, 126684 (2021). https://doi.org/10.1016/j.jtemb.2020.126684 https://doi.org/10.1016/j.jtemb.2020.126684
A. Przybylowicz, P. Chesy, M. Herman, A. Parczewski, S. Walas, and W. Piekoszewski, Cen. Eur. J. Chem., 10(5), 1590-1599 (2012). https://doi.org/10.2478/s11532-012-0089-z https://doi.org/10.2478/s11532-012-0089-z
H. Al-Muzafar and M. Al-Hariri, J. Med. Life., 15(5), 601-605 (2022). https://doi.org/10.25122/jml-2022-0017 https://doi.org/10.25122/jml-2022-0017
P. T. Huynh,T. P. N. Trinh, D. H. Binh, N. T. T. Linh, T. T. H. Loan, T. T. Anh, H. M. Dung, and N. V. Dong, Sci. Technol. Dev., 22(4), 370-377 (2020). https://doi.org/10.32508/stdj.v22i4.1698 https://doi.org/10.32508/stdj.v22i4.1698
D. E. B. Fleming, M. N. Nader, K. A. Foran, C. Groskopf, M. C. Reno, C. S. Ware, M. Tehrani, D. Guimarães, and P. J. Parsons, Appl. Radiat. Isot., 120, 1-6 (2017). https://doi.org/10.1016/j.apradiso.2016.11.015 https://doi.org/10.1016/j.apradiso.2016.11.015
S. O. Olabanji, O. A. Ajose, N. O. Makinde, M. C. Buoso, D. Ceccato, M. De Poli, and G. Moschini, Nucl. Instrum. Methods, 240(4), 95-907 (2005). https://doi.org/10.1016/j.nimb.2005.06.206 https://doi.org/10.1016/j.nimb.2005.06.206
M. A. Nelson, L. A. Triplett, J. J. Colman, and R. RousselDupré, Nucl. Instrum. Methods, 597(1), 110-114 (2008). https://doi.org/10.1016/j.nima.2008.08.059 https://doi.org/10.1016/j.nima.2008.08.059
D. E. B. Fleming, M. G. Kaiser, B. D. Rankin, and K. M. M. Schenkels, J. Trace Elem. Med. Biol., 77, 127136 (2023). https://doi.org/10.1016/j.jtemb.2023.127136 https://doi.org/10.1016/j.jtemb.2023.127136
K. He, Eur. J. Clin. Invest., 41(1), 98-102 (2011). https://doi.org/10.1111/J.1365-2362.2010.02373.X https://doi.org/10.1111/J.1365-2362.2010.02373
M. Bhatia, A. J. Specht, V. Ramya, D. Sulaiman, M. Konda, P. Balcom, E. M. Sunderland, and A. Qureshi, Environ. Sci. Technol., 55(19), 13113-13121 (2021). https://doi.org/10.1021/acs.est.1c00937 https://doi.org/10.1021/acs.est.1c00937
U. Sharma, K. Van Delinder, J. L. Gräfe, and D. E. B. Fleming, Appl. Radiat. Isot., 182, 110151 (2022). https://doi.org/10.1016/j.apradiso.2022.110151 https://doi.org/10.1016/j.apradiso.2022.110151
D. E. B. Fleming, S. L. Crook, and C. T. Evans, Appl. Radiat. Isot., 145, 170-175 (2019). https://doi.org/10.1016/j.apradiso.2018.12.033 https://doi.org/10.1016/j.apradiso.2018.12.033
D. E. B. Fleming, S. L. Crook, C. T. Evans, M. N. Nader, M. Atia, M. T. Hicks, E. Sweeney, C. R. McFarlane, J. Kim, E. Keltie, and A. Adisesh, J. Trace Elem. Med. Biol., 62, 126603 (2020). https://doi.org/10.1016/j.jtemb.2020.126603 https://doi.org/10.1016/j.jtemb.2020.126603
D. K. T. V. K. Singh and J. Kawai, 'X‐Ray Fluorescence in Biological Sciences: Principles, Instrumentation, and Applications', Wiley, New Jersey, 2022. https://doi.org/10.1002/9781119645719 https://doi.org/10.1002/9781119645719
D. Fleming, S. Bennett, and C. Frederickson. J. Trace Elem. Med. Biol., 50, 609-614 (2018). https://doi.org/10.1016/j.jtemb.2018.04.025 https://doi.org/10.1016/j.jtemb.2018.04.025
D. E. B. Fleming, X-Ray Spec., 51, 328-337 (2022). https://doi.org/10.1002/xrs.3248 https://doi.org/10.1002/xrs.3248
A. Z. Taha and O. A. Merghani, J. Community Health, 15(4), 267-274 (1990). https://doi.org/10.1007/BF01350292 https://doi.org/10.1007/BF01350292
D. E. B. Fleming, S. L. Crook, C. T. Evans, M. N. Nader, M. Atia, J. M. T. Hicks, E. Sweeney, C. R. McFarlane, J. S. Kim, E. Keltie, and A. Adisesh, Appl. Radiat. Isot., 167, 109491 (2021). https://doi.org/10.1016/j.apradiso.2020.109491 https://doi.org/10.1016/j.apradiso.2020.109491
A. M. Cuevas, F. Bernardini, A. Gianoncelli, and C. Tuniz, X-Ray Spectrom., 44(3),105-115 (2015). https://doi.org/10.1002/xrs.2585 https://doi.org/10.1002/xrs.2585
S. O. Olabanji, O. R. Omobuwajo, D. Ceccato, A. C. Adebajo, M. C. Buoso, and G. Moschini, Nucl. Instrum. Methods, 266(10), 2387-2390 (2008). https://doi.org/10.1016/j.nimb.2008.03.016 https://doi.org/10.1016/j.nimb.2008.03.016
G. Mattiello and A. Bortoli, Ann. Ist. Super. San., 31(2), 233-237 (1995). https://doi.org/10.1007/BF00380607 https://doi.org/10.1007/BF00380607
V. A. Trunova, N. V. Brenner, and V. V. Zvereva, Toxicol. Mech. Methods., 19(1) 1-18, (2009). https://doi.org/10.1080/15376510801891310 https://doi.org/10.1080/15376510801891310
D. E. B. Fleming, X-Ray Spectrom., 51, 1-23 (2021). doi: 10.1002/xrs.3248 https://doi.org/10.1002/xrs.3248
Y. Li, X. Zou, J. Lv, L. Yang, H. Li, and W. Wang, Bio. T. Elem. Res., 145(2), 158-165 (2012). https://doi.org/10.1007/s12011-011-9187-6 https://doi.org/10.1007/s12011-011-9187-6
Y. Takasaki, S. Sakagawa, K. Inagaki, S. Fujii, A. Sabarudin, T. Umemura, and H. Haraguchi, Talanta, 87(1), 24-29 (2011). https://doi.org/10.1016/j.talanta.2011.09.022 https://doi.org/10.1016/j.talanta.2011.09.022