
- P-ISSN 1225-0163
- E-ISSN 2288-8985







Glycans, complex carbohydrates essential for numerous biological processes, present significant analytical challenges due to their structural complexity and microheterogeneity. This study introduces 4 aminophenyl ferrocenecarboxamide (APF) as a novel glycan labeling reagent designed to improve the sensitivity, stability, and separability of glycans in analytical workflows. Synthesized through the reaction of N-succinimidyl ferrocenecarboxylate and p-phenylenediamine, APF combines the electrochemical activity of ferrocene with the labeling efficacy of aromatic amines, enhancing hydrophobicity and facilitating separation. APF-labeled biantennary sialyl N-linked glycans were analyzed using liquid chromatography-mass spectrometry (LC-MS), demonstrating increased mass, retention time, and distinct fragmentation patterns that enable detailed structural elucidation. The results highlight APF’s versatility in glycan profiling, offering improved performance across electrochemical, spectroscopic, and mass spectrometric platforms. With potential applications in clinical diagnostics, biomarker discovery, and therapeutic research, APF represents a significant advancement in glycomics, providing a robust and efficient tool for the analysis of complex glycans.
Glycans, complex carbohydrates attached to proteins and lipids, play critical roles in various biological processes, including cell signaling, immune response, and protein folding.1,2 Due to their structural complexity and microheterogeneity, glycans pose significant challenges in analysis, requiring sophisticated techniques for their accurate identification and characterization. To address these challenges, derivatization methods are commonly employed to enhance the detectability, stability, and separability of glycans in analytical procedures such as liquid chromatography (LC), mass spectrometry (MS), and spectroscopic analysis.3,4 Conventional glycan labeling strategies typically employ fluorescent or isotopic tags to facilitate separation, detection, and quantification.5-8 Therefore, there is a continuous need for developing new and more versatile reagents for glycan labeling that offer enhanced performance across multiple analytical platforms.
Ferrocene-based reagents have gained attention in recent years due to their unique properties, such as high stability, electrochemical activity, and the ability to facilitate improved ionization in mass spectrometry.9,10 These reagents offer advantages for a range of applications, including electrochemical sensing, atomic and molecular spectroscopy, and mass spectrometry coupled with separation techniques.11-15
In this study, the development of 4-aminophenyl ferrocenecarboxamide (APF) as a glycan labeling reagent represents a promising advance. By combining the ferrocene moiety with an aromatic amine, APF not only improves the electrochemical and spectroscopic properties of glycans but also enhances their hydrophobicity, which aids in separation and purification processes. We demonstrate that APF effectively labels glycans, offering significant improvements in sensitivity, separation efficiency, and structural analysis when analyzed by LC-MS/MS. This research shows the application of APF to biantennary sialyl N-linked glycans derived from glycopeptides.
By addressing limitations in existing glycan labeling techniques, this study establishes APF as a robust reagent that enhances the analytical workflow for the complex glycans. Glycan labeling remains a cornerstone of glycomics research, enabling detailed structural analysis and functional understanding of these critical biomolecules. The novel APF-based labeling strategy will offer significant advancements in glycan profiling, with potential applications extending to clinical diagnostics, biomarker discovery, and therapeutic research .
The peptide:N-glycosidase F (PNGase F) was purchased from BioLabs (Ipswich, MA). The sialylglycopeptide (SGP, > 95 %) was purchased from FUSHIMI (Kagawa, Japan). The N-succinimidyl ferrocenecarboxylate was purchased from TCI (Japan). The cation exchange resin was purchased from The Nest Group (Southborough, MA, USA). All other reagents and solvents such as p-phenylenediamine, dimethylformamide (DMF), sodium cyanoborohydride (NaBH3CN), hydrochloric acid (HCl), acetic acid, dimethyl sulfoxide (DMSO), anhydrous methanol, 50 % sodium hydroxide (NaOH), iodomethane (CH3I), milli-Q-water, dichloromethane (DCM), formic acid, and acetonitrile (ACN) were purchased from SigmaAldrich (St. Louis, Missouri, US).
The reaction of N-succinimidyl ferrocenecarboxylate with p-phenylenediamine is a conjugation reaction that forms a stable amide bond between carboxylate and amine. 100 mM N-succinimidyl ferrocenecarboxylate and 2 M p-phenylenediamine were dissolved in DMF to prepare stock solutions, respectively. Nsuccinimidyl ferrocenecarboxylate and p-phenylenediamine were transferred to a glass tube in an equivalent ratio of 1:20, mixed, and reacted. The reaction was performed at room temperature with vortexing every 20 min for 2 h. The purification of 4-aminophenyl ferrocenecarboxamide (APF) was performed by liquid-liquid extraction in which DCM was added to the reaction solution and then a 0.1 % formic acid aqueous solution was added and removed several times to remove the solvent and excess p-phenylenediamine.
From the sialylglycopeptide (SGP) stock (2 mg/ mL) dissolved in water, 20 mg of SGP was taken into an Eppendorf tube and dried. After dissolving SGP in 1 × glycoprotein denaturing buffer, 1 munits of PNGase F was added and digested overnight at 37 °C to release biantennary sialic N-linked glycans. The solution was purified to the glycans through cation exchange and dried.
The biantennary sialic N-linked glycans were conjugated with 4-aminophenyl ferrocenecarboxamide using a reductive amination. Permethylation of labeled glycans is as follows. The labeled glycans were resuspended in 200 µL dimethyl sulfoxide (DMSO) and 250 µL freshly dehydrated base (50 % NaOH in 2 mL of anhydrous DMSO). After sonication and vortexing under nitrogen, 100 µL iodomethane (CH3I) was added and the mixture was vortexed vigorously for 10 min. 2 mL Milli-Q water was added to the labeled glycan sample and excess CH3I was removed by bubbling with a stream of nitrogen gas. After bubbling, the salt and remaining DMSO were removed by liquid-liquid extraction with water and DCM. The permethylated labeled glycans in DCM were dried under a nitrogen stream and stored at -20 °C for mass spectrometry analysis.
The labeled glycans and synthesized labeling reagent were jointly analyzed using an LCQ Fleet mass spectrometer (Thermo Fisher Scientific, USA) at the Gyeongnam Bio and Anti-aging Core Facility Center for CWNU and the Chronic and Metabolic Diseases Research Center, Sookmyung Women’s University, equipped with an Accela 600 pump, Nano-flow Tee Splitter, and nano-electrospray ionization system. The glycan sample was resuspended in 50 µL of buffer B (0.1 % formic acid in 80 % acetonitrile) solution and 5 µL of the sample was injected for nanoLC-MS. Glycans were separated with a gradient of buffer A (0.1 % formic acid) and buffer B and ionization was performed by Nanospray Ion Source with a selfpacked C18 capillary column of a PicoTipTM emitter (PicoFritTM SELF/P, 360 µm OD × 75 µm ID × 15 µm, New Objective) at 2.5 kV capillary voltage and 200 capillary temperature. Full MS spectra of glycans were collected in the mass range 150 ~ 2,000 m/z in positive ion and profile mode. MS/MS spectra of top 5 datadependent scans were obtained in centroid mode with settings of collision energy 38 %, isolation width 2 Da, and activation time 10 ms.
Ferrocene-based derivatization is widely used in various fields of analytical chemistry, such as electrochemistry, atomic/molecular spectroscopy, and mass spectrometry combined with separation methods. The development of glycan derivatives with ferrocene is expected to provide various tools for the analysis of glycans. For this reason, 4-aminophenyl ferrocenecarboxamide (APF) was synthesized as a glycan labeling reagent for reductive amination by conjugation reaction of N-succinimidyl ferrocenecarboxylate and p-phenylenediamine as shown in Fig. 1.
Fig. 1(a) shows the synthesis of 4-aminophenyl ferrocenecarboxamide by nucleophilic attack of p-phenylenediamine on N-succinimidyl ferrocenecarboxylate, and its theoretical mass is shown to be 321.0690 Da (mono). After purification, the reagent was analyzed by LC-MS and was measured as [M+H]+ = 321.18 Da, indicating successful synthesis in Fig. 1(b). Fig. 1(c) shows the MS/MS spectrum of y ion fragments showing the cleavage of carbonyl-ferrocene bond of 186 Da and amide bond of 213 Da in the synthetic reagent.
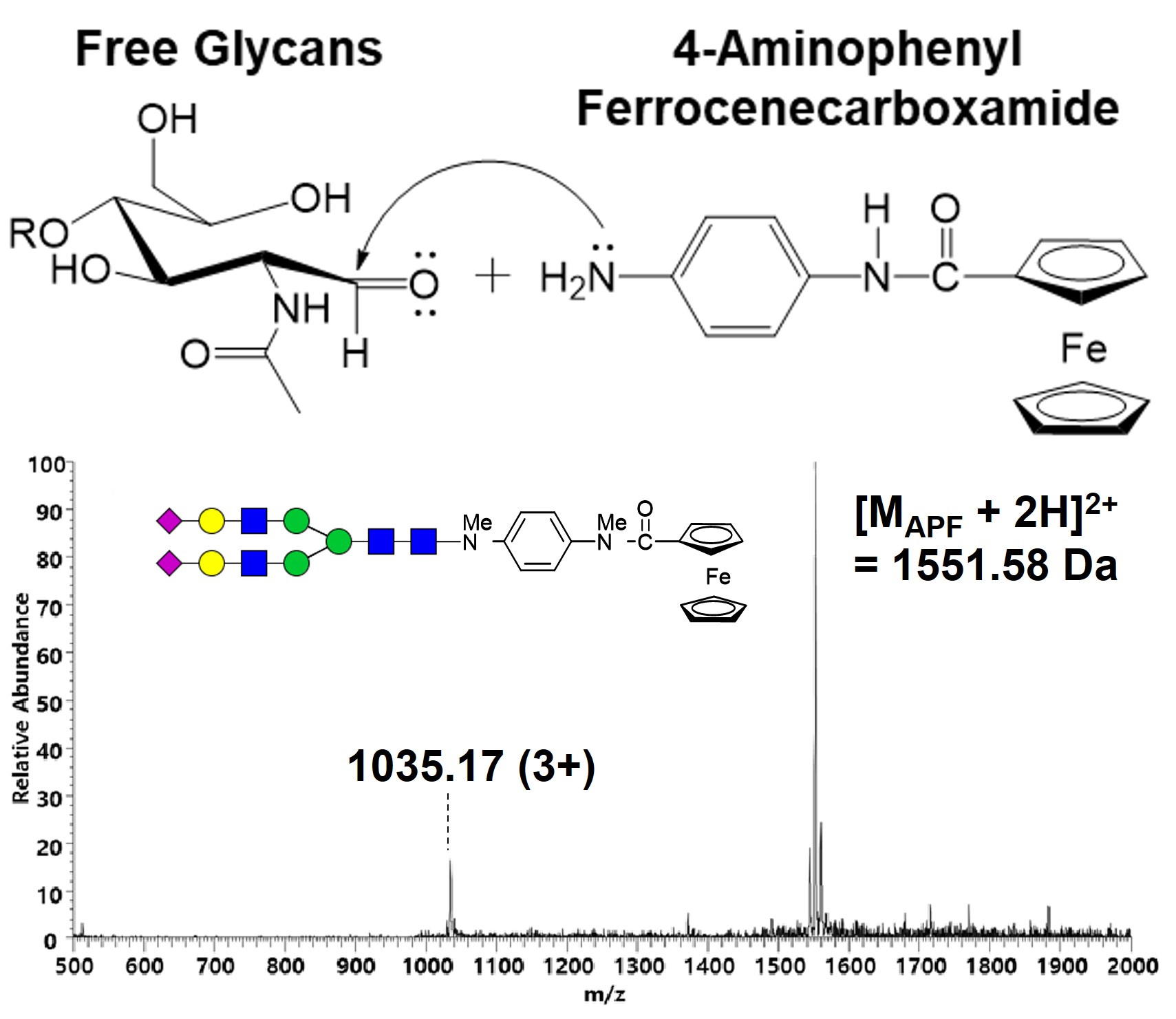
Various aromatic amines are commonly used as fluorescent derivatives of glycans for LC analysis and quantitative mass spectrometry by isotope-labeled derivatives of these. This approach is the derivatization of glycans by reductive amination using a newly synthesized ferrocene derivative, 4-aminophenyl ferrocenecarboxamide. Fig. 2 shows glycan labeling by reductive amination by conjugating the amine of 4-aminophenyl ferrocenecarboxamide to the aldehyde of the reducing end of free glycans to form a Schiff base and reducing it with a reducing agent (NaBH3CN). The labeled glycans were purified by liquid-liquid extraction and prepared for mass spectrometry through permethylation for chemical stability and ionization efficiency.
Glycan labeling reagent, 4-aminophenyl ferrocenecarboxamide, is expected to be utilized as a versatile tool in glycan analysis. 4-Aminophenyl ferrocenecarboxamide contains both ferrocene and aromatic groups, which may contribute to its potential as a fluorophore or chromophore. Due to these structural features, it is being investigated for possible applications in areas such as electrochemistry, atomic analysis, and mass spectrometry. Additionally, this reagent is expected to be useful for purifying and separating glycans of various hydrophilic structures due to increased hydrophobicity.
Free biantennary sialic N-linked glycan released from SGP was labeled with 4-aminophenyl ferrocenecarboxamide. Unlabeled or labeled biantennary sialic N-linked glycans were separated by liquid chromatography and analyzed by mass spectrometry. Glycans were eluted from high buffer B by the gradient, and unlabeled glycans were eluted at 58 min and labeled glycans at 63 min, respectively, showing separation with a retention time of 5 min due to hydrophobicity. Fig. 3 shows the full mass spectra of unlabeled and labeled glycans. Glycans were analyzed as charge states of both divalent and trivalent cations by protonation. As shown in Fig. 3(a) and (b), the mass spectrum of the unlabeled glycan and the labeled glycan was obtained with a mass of [M +2H]2+ = 1385.50 Da and [M +2H]2+ = 1551.58 Da, respectively. The mass spectrum of APF-labeled glycans shows that APF is stably labeled and detected on glycans during purification, permethylation, and ionization for mass spectrometry.
Fig. 4 shows the MS/MS spectrum of 1551.58 Da (2+) for structural interpretation of glycans by fragmentation of APF-labeled glycans. In the fragmentation pattern, APF is structurally rigid, while the fragmentation of glycans is rich to facilitate structural interpretation of glycans. In general, dissociation of glycans appears as doubly charged ions, and dissociation of APF and glycans shows rich fragmentation from singly charged ions.
The development of 4-aminophenyl ferrocenecarboxamide (APF) as a glycan labeling reagent represents a significant advancement in the tools available for glycan analysis. Expected to open new possibilities for glycomics research, APF is an ideal glycan labeling reagent for high-throughput glycan analysis for both research and clinical applications. The versatility of 4-aminophenyl ferrocenecarboxamide is highlighted by its potential use in a wide range of glycan-related applications. Beyond mass spectrometry, the ferrocene component of the reagent suggests potential applications in electrochemistry and fluorescence-based assays, where the electrochemical properties of ferrocene could be exploited for sensitive detection. Furthermore, the ability of APF to enhance glycan separation through increased hydrophobicity positions it as a valuable tool for glycan purification and fractionation, which are important steps in the study of complex glycan mixtures and their functional roles in biology.
The synthesis of APF with N-succinimidyl ferrocenecarboxylate and p-phenylenediamine was successfully performed, and the product was characterized using mass spectrometry. The full and MS/MS mass spectra for APF confirmed the successful synthesis of the expected product with a high degree of purity, which is essential for reliable labeling reactions. APF by synthetic mechanism with proposed reagents provides a reproducible and efficient approach for glycan labeling, which is important for glycan analysis. The labeling of glycans with APF via reductive amination was shown to be effective, producing labeled glycans suitable for analysis by mass spectrometry.
The application of LC-MS to analyze the labeled glycans revealed clear differences in the mass spectra of unlabeled and APF-labeled glycans, with the labeled glycans showing an increase in mass corresponding to the addition of the APF tag. The MS/MS analysis of the labeled glycans further provided valuable structural insights, with clear fragmentation patterns that allowed for detailed interpretation of glycan structures. The APF label itself remained stable during the fragmentation process, enabling a more accurate analysis of glycan structure by minimizing the potential for fragmentation of the label. The versatility of 4-aminophenyl ferrocenecarboxamide is highlighted by its potential use in a wide range of glycan-related applications. The ability of APF to enhance glycan separation through increased hydrophobicity positions it as a valuable tool for glycan purification and fractionation, which are important steps in the study of complex glycan mixtures and their functional roles in biology.
Future work should also explore the compatibility of APF with a broader range of glycan types, including glycans released from various glycoproteins. Furthermore, the application of APF in high-throughput screening for glycan biomarkers holds great promise for clinical diagnostics and therapeutic development.
M. He, X. Zhou, and X. Wang, Signal Transduct. Target. Ther., 9(194) (2024). https://doi.org/10.1038/s41392-024-01886-1 https://doi.org/10.1038/s41392-024-01886-1
D. Chettri, M. Chirania, D. Boro, and A. K. Verma, Life Sci., 348, 122689 (2024). https://doi.org/10.1016/j.lfs.2024.122689 https://doi.org/10.1016/j.lfs.2024.122689
D. G. Delafield and L. Li, Mol. Cell Proteomics., 20, 100054 (2021). 10.1074/mcp.R120.002095 https://doi.org/10.1074/mcp.R120.002095
A. Shubhakar, K. R. Reiding, R. A. Gardner, D. I. Spencer, D. L. Fernandes, and M. Wuhrer, Chromatographia., 78(5-6), 321-333 (2015). https://doi.org/10.1007/s10337-014-2803-9 https://doi.org/10.1007/s10337-014-2803-9
Y. Helali and C. Delporte, J. Chromatogr. B, 1237, 124068 (2024). https://doi.org/10.1016/j.jchromb.2024. https://doi.org/10.1016/j.jchromb.2024
Z. L. Wu and J. M. Ertelt, Commun. Biol., 5, 501 (2022). https://doi.org/10.1038/s42003-022-03444-3 https://doi.org/10.1038/s42003-022-03444-3
Z. L. Wu, A. D. Person, A. J. Burton, R. Singh, B. Burroughs, D. Fryxell, T. J. Tatge, and T. Manning, G. Wu, K. A. D. Swift, and V. Kalabokis, Glycobiology, 29(11), 750-754 (2019). https://doi.org/10.1093/glycob/cwz058 https://doi.org/10.1093/glycob/cwz058
J. Yun, J. Y. Jo, S. T. Tuomivaara, and J.M. Lim, Microchem. J., 170, 106655 (2021). https://doi.org/10.1016/j.microc.2021.106655 https://doi.org/10.1016/j.microc.2021.106655
U. Rauf, G. Shabir, S. Bukhari, F. Albericio, and A. Saeed, Molecules, 28, 5765 (2023). https://doi.org/10.3390/molecules28155765 https://doi.org/10.3390/molecules28155765
B. Seiwert and U. Karst, Anal. Bioanal. Chem., 390, 181-200 (2008). https://doi.org/10.1007/s00216-007-1639-7 https://doi.org/10.1007/s00216-007-1639-7
I. Alshanski, Y. Sukhran, E. Mervinetsky, C. Unverzagt, S. Yitzchaik, and M. Hurevich, Biosens Bioelectron., 172, 112762 (2021). https://doi.org/10.1016/j.bios.2020.112762 https://doi.org/10.1016/j.bios.2020.112762
I. S. Butler, R. P. Kengne-Momo, A. Vessières, G. Jaouen, and C. Policar, Appl. Spectrosc. Rev., 47(8), 620-632 (2012). https://doi.org/10.1080/05704928.2012.697088 https://doi.org/10.1080/05704928.2012.697088
J. Bariyanga, J. Mol. Struct., 657, 225-237 (2003). https://doi.org/10.1016/S0022-2860(03)00406-X https://doi.org/10.1016/S0022-2860(03)00406-
J. M. Quirke and G. J. Van Berkel, J. Mass Spectrom., 36(2), 179-87 (2001). https://doi.org/10.1002/jms.119 https://doi.org/10.1002/jms.119
Y. Okada and M. Tsuchida, Int. J. Anal. Mass Spectrom. Chromatogr., 7, 1-8 (2019). https://doi.org/10.4236/ijamsc.2019.71001 https://doi.org/10.4236/ijamsc.2019.71001