
- P-ISSN 1225-0163
- E-ISSN 2288-8985



Edible insects have gained considerable attention as sustainable and nutrient-rich protein sources in the food industry. This review summarizes key advancements in edible insect protein and oil extraction techniques. Efficient protein recovery techniques are discussed. In addition, strategies such as defatting (which enhances extraction efficiency) are also discussed. The nutritional significance of edible insect-derived peptides, amino acids, and fatty acids is also reviewed, with emphasis on their potential as food ingredients. Analytical techniques for characterizing edible insect volatile compounds are described, along with the key identified volatiles. Furthermore, fermentation has been explored as a strategy to enhance the consumer acceptance of edible insects. These insights support the development of innovative and sustainable food products that utilize edible insect resources.
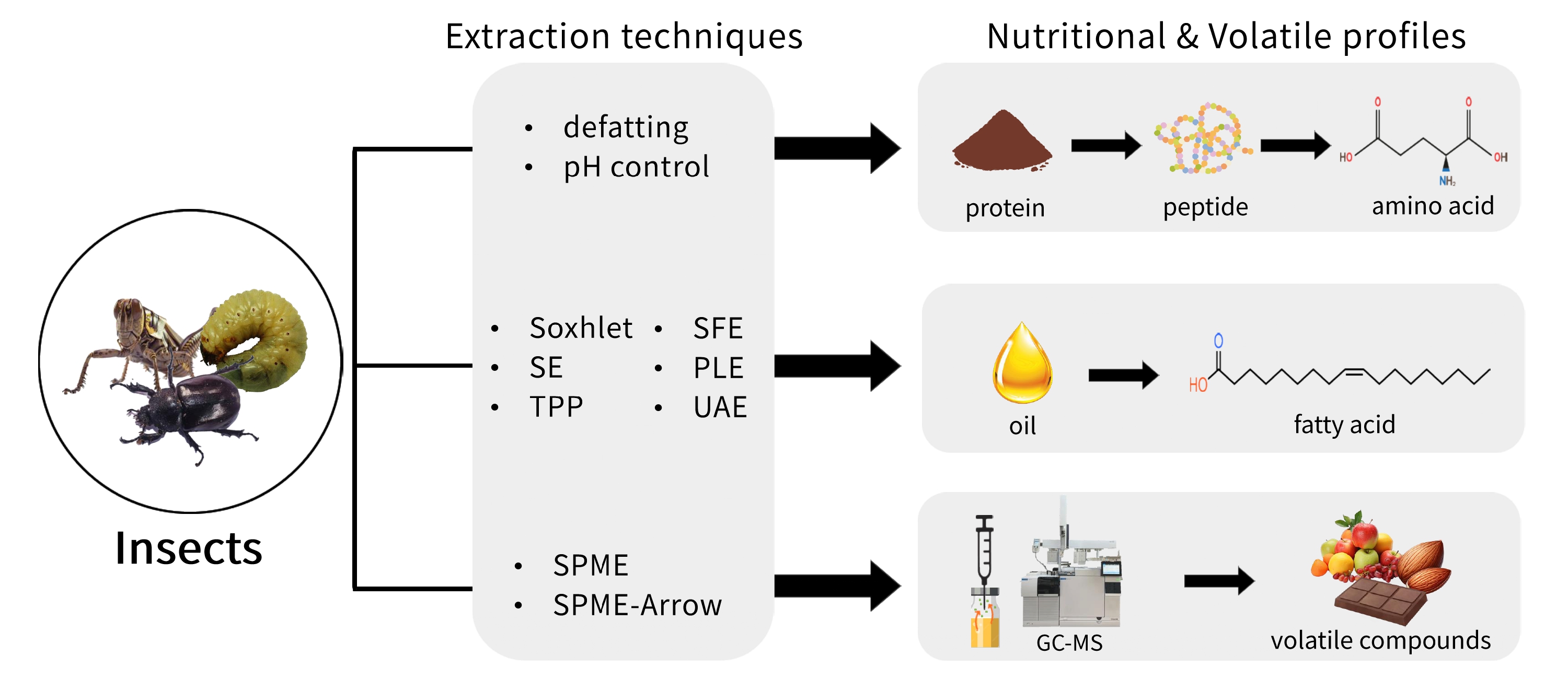
Edible insects have garnered significant research interest because of their uses in various sectors, including the food industry.1 Compared to conventional livestock, they require fewer resources, such as water and land, while emitting lower levels of CO2 and ammonia.2 Consequently, edible insects are regarded as a promising alternative food source for achieving the United Nations (UN) Sustainable Development Goals (SDGs). As the global population is projected to reach approximately nine billion by 2050, concerns regarding the sufficiency of food supply continue to rise.3 The Food and Agriculture Organization (FAO) has recognized edible insects as a sustainable and nutritious food source for human consumption.4 Currently, more than 2,100 species of edible insects are consumed by approximately two billion people across more than 113 countries.5 As the market for edible insects continues to expand, they are increasingly being recognized as a valuable food source.
Currently, thirty-five African countries, Mexico, China, Thailand, and India, have been identified as major consumers of edible insects, demonstrating the greatest levels of species diversity. Furthermore, twentynine countries in Asia, twenty-three in the Americas, fourteen in Oceania, and eleven in Europe have been documented as regions where edible insects are widely consumed.6 Beetles (Coleoptera, 31 %) are the most commonly consumed edible insects, followed by caterpillars (Lepidoptera, 18 %), bees, wasps, and ants (Hymenoptera, 14 %), and grasshoppers, locusts, and crickets (Orthoptera, 13 %). Other consumed groups include Hemiptera (10 %), termites (Isoptera, 3 %), dragonflies (Odonata, 3 %), flies (Diptera, 2 %), and various others (5 %).6 The European Union (EU) maintains strict regulatory frameworks regarding the use of edible insects.7 Nevertheless, research is ongoing on their potential as food and pet feed, especially using Hermetia illucens, Tenebrio molitor, and Bombyx mori.6 In Croatia and Italy, maggot cheese has traditionally been regarded as a delicacy. In several African countries, edible insect consumption primarily relies on traditional harvesting from natural habitats. Across Asia, edible insects with reported anticancer and immunomodulatory effects are being utilized in the development of functional food products.8
The bioavailability of edible insect-derived proteins is comparable to that of soybeans, indicating that they are readily digested and absorbed in the human body.9 In addition, they aid various biological activities, including anti-inflammatory effects, alleviation of abdominal pain, and immune system enhancement.9,10 Edible insect-derived proteins are being actively explored for additional functional properties. Furthermore, insect oil is obtained as a by-product of the defatting process employed for protein extraction.11 Edible insect oils are a good source of essential fatty acids, such as α-linolenic acid and linoleic acid.2 Therefore, oil derived from edible insects has the potential to contribute to the development of high value-added technologies and serve as a valuable upcycling resource.
Volatile compounds are key contributors to food quality, as they enhance the sensory experience of eating.12 Hence, the volatile profile of foods significantly influences consumer acceptance.13,14 Therefore, to promote the utilization of edible insects in the food industry, the establishment a comprehensive dataset on their volatile compound profiles is essential. Moreover, the impact of processing methods such as fermentation on volatile compounds should be analyzed, given that fermentation is widely used to improve both the volatile profile and safety of foods.15 Therefore, this review investigates the key volatile compounds in edible insects and the changes in their volatile profiles resulting from fermentation.
The growing interest in edible insects has inspired research on them across various fields. However, comprehensive literature detailing the extraction methods, nutritional composition, and volatile profiles of edible insects is limited. This review highlights the potential of edible insects as a sustainable food source, covering the various extraction methods for proteins and oils. Additionally, the nutritional composition of amino acids and fatty acids, along with key volatile compounds and the effects of fermentation, is also presented.
Edible insects are recognized as a rich source of dietary protein, with protein content ranging from 31.06 % to 71.04 %.9 Efficient protein extraction from edible insects is highly dependent on the choice of extraction method, with factors such as solvent type and intrinsic protein characteristics playing key roles.16 Selecting an appropriate extraction method is crucial for optimizing protein yield and functionality.17-19 The protein yields obtained using different extraction techniques are presented in Table 1.
Defatting is a common step in edible insect protein extraction and is typically performed using organic solvents to enhance the efficiency. This process has been shown to improve protein yield, as demonstrated in Protaetia brevitarsis larvae, where defatting increased the protein yield from 68.77 % to 77.62 %.20 Hexane is a widely used degreasing solvent, effectively removing over 96 % of oil and enhancing the functional properties of edible insect proteins.21,22 Hexane has demonstrated superior protein extraction efficiency, achieving a protein yield of 77.62 %, compared to methanol (70.41 %) and ethanol (68.39 %).22 After degreasing, edible insect-derived proteins are typically extracted using pH-based solutions such as NaOH and distilled water.17-22 Protein yields achieved by adjusting the pH using NaOH in various edible insect species were reported as follows: 49.90 % for T. molitor, 62.32 % for Allomyrina dichotoma larvae, 61.23 % for Acheta domesticus, and 58.17 % for Locusta migratoria. 17
In contrast to the conventional methods, some studies have reported successful protein extraction without pH adjustment. Hexane was utilized to defat T. molitor, A. dichotoma, and P. brevitarsis, followed by the rapid extraction of proteins using 0.02 % (w/v) ascorbic acid and 0.58 M saline.16 Sonication has been reported to enhance protein yields. The protein yield of Schistocerca gregaria extracted using NaOH was 52.9 %; however, it increased to 57.50 % using sonication. Similarly, the protein yield of Apis mellifera from sonification (55.20 %) was higher compared to that from NaOH extraction (39.60 %). This highlights that improved protein recovery is achieved when sonication is used instead of solvent-based extraction methods.19
Ethanol treatment during protein extraction enhanced the functional properties of the proteins. The freezedried T. molitor larval powder was initially defatted using hexane and subsequently dissolved in distilled water to achieve a concentration of 10 %. The pH was adjusted using NaOH, and ethanol was added to the supernatant. This treatment facilitated the breakdown of high-molecular-weight proteins into low-molecularweight proteins, accompanied by structural modifications such as a reduction in the proportion of α-helices and an increase in β-sheets.18
These results highlight the importance of selecting appropriate processing methods for optimizing protein recovery. Protein yield varies significantly depending on the edible insect species and processing conditions applied. For example, the effectiveness of pH adjustment and solvent selection varies between species: NaOH treatment yielded 49.90 % in T. molitor and 62.32 % in Alphitobius diaperinus larvae.17 These differences emphasize that for maximizing the protein yield, appropriate extraction methods must be selected, depending on the characteristics of each edible insect species.
In edible insects, peptides are the basic units that constitute the proteins. Sequence analysis is essential to understand the structural and functional properties of edible insect proteins. However, studies on edible insect-derived peptides are limited due to structural changes caused by thermal processes during sterilization and storage.23 Consequently, establishing a comprehensive database of edible insect-derived peptides is essential to support further research.
Peptide analysis is an important approach for identifying edible insect species and exploring their functional properties. Peptide profiling of A. domesticus was performed using ultra-performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry (UHPLC-QTOF-MS/MS). Four peptide sequences specific to the Acheta genus were identified: AAELSGDAQTAVR, QQFPDGAQAADK, VQEAVQPHADAVAESLK, and LSNHLSNLFK. These peptides are considered potential authentication markers for the identification of A. domesticus.24 Furthermore, peptides with angiotensin-converting enzyme (ACE) inhibitory activity (which contribute to blood pressure regulation) have been identified in T. molitor larvae. Reversed-phase high-performance liquid chromatography (RP-HPLC) analysis revealed peptide sequences such as Tyr-Ala-Asn, YAN, NIKY, QGLGY, and HILG.25,26 In addition, four ACE inhibitory peptides (SY, PF, YPY, and WI) were identified in P. brevitarsis larvae.27 These peptides promote vasodilation and help reduce blood pressure.25
The amino acid profile offers valuable insights into the nutritional value, protein conversion efficiency, and potential functional properties of edible insect proteins.22 Amino acid analysis of insects is conducted using an amino acid analyzer. The amino acid content varies based on the preprocessing methods, such as freezedrying and defatting. The amino acid compositions of the edible insects are listed in Table 2.
The major amino acids identified in A. domesticus were alanine (80.52 – 222.21 mg/g), glutamic acid (37.72 – 99.51 mg/g), and glycine (47.21 – 93.73 mg/g). A. domesticus contains various essential amino acids (EAAs) such as proline and aspartic acid, along with other amino acids vital for human health.28,29
In the case of H. illucens, the predominant amino acids identified were glutamic acid (0.11 – 1.07 mg/g), aspartic acid (0.12 – 1.19 mg/g), and leucine (0.84 – 0.87 mg/g), along with methionine, cysteine, and tryptophan.30,31 The FAO has reported that the recommended intake of sulfur-containing amino acids (in particular methionine and cysteine) is 2.30 % for children, adolescents, and adults.32 Therefore, H. illucens is a suitable dietary source of sulfur-containing amino acids.30
The amino acids in freeze-dried P. brevitarsis larvae showed the highest contents of glutamic acid (10.17 – 73.88 mg/g), aspartic acid (4.98 – 39.55 mg/g), and proline (12.18 – 184.33 mg/g). In addition, EAAs, including histidine, isoleucine, and leucine, have also been identified.22,33 These EAAs are not capable of being synthesized by the human body; therefore, their intake is crucial for proper nutrition and is strongly recommended.35 Similarly, the major amino acids identified in A. dichotoma and B. mori are glutamic acid, aspartic acid, and proline.13,33-35
The major amino acids identified in freeze-dried T. molitor larvae are proline (6.59 – 80.30 mg/g), glutamic acid (10.37 – 44.64 mg/g), and phenylalanine (7.93 – 13.41 mg/g). Moreover, it includes several essential amino acids such as lysine and valine, which are important for human health.13,33
Edible insects contain high levels of glutamic acid (0.11 – 495.00 mg/g) and aspartic acid (0.12 – 91.00 mg/g). These values are comparable to those in beef, with glutamic acid at 75.10 mg/g and aspartic acid at 41.10 mg/g.36 Similar levels have also been reported in fish (pomfrets), which contains 114.00 mg/g of glutamic acid and 76.00 mg/g of aspartic acid.37 The edible insect protein has a well-balanced amino acid profile, with EAAs such as lysine, which are not available in grains, tubers, and legumes.38
Moreover, amino acids contribute to the development of tastes such as umami, bitterness, and sweetness, thereby significantly influencing the sensory properties of food.39 Umami provides a savory sensation and enhances the overall intensity of other tastes.40 Among the amino acids contributing to umami taste, glutamic acid and aspartic acid have been identified as predominant in edible insects, similar to their abundance in beef, pomfrets, and soybeans.30,31,36.39 Methionine, an amino acid known to be associated with bitter taste, is abundant in soybeans but only present in trace amounts in edible insects.41 Methionine content could potentially contribute to differences in bitterness between soybeans and edible insects. Therefore, edible insects are considered to exhibit sensory properties comparable to those of conventional protein sources such as meat, fish, and soybeans.
Hydrophobic amino acids, valine, alanine, isoleucine, and leucine, facilitate antioxidant activities.42 Furthermore, leucine, arginine, phenylalanine, proline, and alanine are responsible for hypoglycemic activities.43 In addition, glutamic acid, alanine, isoleucine, tryptophan, methionine, phenylalanine, proline, and valine are important amino acids for facilitating lipid metabolism.44 In particular, glutamic acid plays a crucial role in nitrogen metabolism, as it participates in nitrogen assimilation and serves as a key component in aminotransferase reactions.45 Aspartic acid contributes to the production of EAAs, such as lysine, threonine, methionine, and isoleucine, through metabolic pathways.43 Hence, insufficient levels of these EAAs are associated with a potential increase in the risk of obesity, cardiovascular diseases, headaches, immune disorders, and other related health conditions.46 Accordingly, edible insects rich in balanced amino acids offer a valuable dietary resource and hold potential in supporting human nutrition.
Lipids account for the second-largest portion of the nutritional composition of edible insects, with a higher content observed during the larval stage.5 The larvae of T. molitor are extensively studied edible insects. Techniques such as Soxhlet extraction, solvent extraction (SE), three-phase partitioning (TPP), supercritical fluid extraction (SFE), pressurized liquid extraction (PLE), and ultrasound-assisted extraction (UAE) have been employed to extract lipids from edible insects. The oil content of T. molitor larvae ranges from 25.43 to 41.72 % (Table 3). The yield of edible insect oil is dependent on the extraction method used. A detailed explanation of each method is provided in this section.
The Soxhlet method has demonstrated the highest oil extraction efficiency (41.72 %) compared to other techniques.47 However, this conventional method has certain limitations, such as requiring a large volume of solvent and involving prolonged extraction at high temperatures.22 Similarly, the n-hexane-based SE method achieves relatively high yields (29.24 %) and is widely employed in industrial oil extraction due to its high oil selectivity, ease of evaporation, and low energy demands.11 However, concerns regarding human health and environmental safety have been raised during the extraction process.48
The TPP technique induces phase separation based on ammonium sulfate concentration, forming three layers: an oil-containing organic phase at the top, a protein-rich layer in the middle, and a hydrophilic layer with polysaccharides at the bottom.47 Although TPP yields less oil than other methods (24.62 %), it efficiently recovers valuable by-products such as proteins, enhancing its utility. SFE is widely used to extract natural extracts and oils. Supercritical CO2, with its optimal critical temperature (31.1 ℃), prevents thermosensitive compound degradation during extraction.24 This non-toxic, easily removable method is environmentally sustainable due to CO2 recovery and reuse.2
The extraction yield of oils is dependent on their solubility in the selected solvent.2 Therefore, to optimize the extraction process, the selection of the extraction solvent should be considered along with the extraction method. A comparison of the oil yields from T. molitor larvae using two ethanol-based extraction methods (PLE and UAE) has been reported.49 The ethanol-based PLE and UAE methods yielded 32.37 % and 28.85 % oil, respectively, both exhibiting high extraction efficiency. PLE is an eco-friendly method that enhances the extraction efficiency by operating at high pressures; this reduces the solvent use and improves the efficiency by decreasing polarity, viscosity, and surface tension.50,51 UAE is another efficient method that minimizes time and energy consumption. By utilizing acoustic cavitation, UAE enhances cell disruption, thereby improving mass transfer and accelerating compound release.52
These findings indicate that edible insects contain considerable amounts of oil extractable through various methods. However, to effectively compare the various extraction methods, it is crucial to consider the edible insect species and the preparation steps performed before oil extraction, such as drying or grinding.
The fatty acid profiles of edible insects depend on their diet, sex, and species. Analysis of edible insect fatty acids is commonly performed using gas chromatography coupled with a flame ionization detector (GC-FID). Oleic acid (C18:1) and linoleic acid (C18:2) were the predominant fatty acids identified in several edible insect species, including T. molitor larvae, Gryllus bimaculatus, and A. domesticus (Table 4). These fatty acids are predominantly identified in most edible insects.2,11,47,53-55
Oleic acid (C18:1), a monounsaturated fatty acid (MUFA) commonly found in olive oil, is recognized for its ability to regulate immune function.56 It increases the beneficial high-density lipoprotein (HDL) cholesterol concentrations in the blood.49 The oleic acid content of T. molitor larvae (42.11 – 44.55 %) was comparable to that of sesame seed oil (43.00 %), rice bran oil (43.42 %), and palmolein (43.36 %).57,58 In contrast, G. bimaculatus showed an oleic acid content (25.46 – 32.15 %) similar to that of pumpkin seed oil (28.70 %) and sunflower seed oil (29.50 %),58 while that of A. domesticus (24.28 – 26.30 %) closely matched with that of soybean oil (24.77 %) and sunflower oil (25.92 %).57 Linoleic acid (C18:2), a type of polyunsaturated fatty acid (PUFA), is primarily present in oils such as safflower, sunflower, and soybean.57 It also acts as a precursor to arachidonic acid (C20:4), which is crucial for the proper functioning of cells and muscles.59 α-Linolenic acid (C18:3), a type of PUFA, is predominantly found in flaxseed, canola, and soybean oils.60 It has diverse health benefits, including cardiovascular protection, anti-obesity, antidiabetic, anti-inflammatory, anticancer, and antioxidant properties.61 Essential fatty acids, such as linoleic acid (an omega-6 fatty acid) and α-linolenic acid (an omega3 fatty acid), must be obtained through the diet since the human body cannot produce them. As shown in Table 4, edible insects contain high levels of linoleic acid (24.29 – 32.82 %) and α-linolenic acid (0.88 – 1.50 %), indicating their potential as a valuable source of essential fatty acids. In particular, the αlinolenic acid contents of the three edible insect species were higher than those found in commonly consumed edible oils, including sesame seed, sunflower seed, pumpkin seed, safflower, and rice bran oils.57,58
Among the saturated fatty acids (SFAs), palmitic acid (C16:0) and stearic acid (C18:0) were identified as the predominant components. These fatty acids have also been identified as the predominant SFAs in beef tallow and lard.62,63 Palmitic acid is a key SFA involved in the structural integrity of cellular membranes and the formation of secretory and transport lipids.64 In addition to its cholesterol-lowering effects on blood lipid profiles, stearic acid has been reported to exert neuroprotective effects against both oxidative stress and excitotoxic neuronal damage.65
Furthermore, the polyunsaturated fatty acid to saturated fatty acid (PUFA/SFA) ratio is a key indicator of the dietary impact on cardiovascular health, with higher ratios suggesting greater nutritional value.66 The PUFA/SFA ratio in Table 4 ranged from 0.75 to 1.31, exceeding the recommended minimum ratio of 0.45 for a healthy human diet. Indicators of coronary artery disease risk, such as the arteriosclerosis index (AI) and thrombosis index (TI) (lower values correspond to improved health outcomes), were found to be below 1 in oils derived from T. molitor larvae, G. bimaculatus, and A. domesticus. 2,54,55 Therefore, edible insect oil is a beneficial source for nutritional intake.
Volatile compounds contribute significantly to food quality through their effects on volatile perception.67 In edible insect studies, several techniques have been applied for the extraction of volatile compounds, including direct solvent extraction, solvent-assisted flavor evaporation (SAFE), and solid-phase microextraction (SPME).2,68,69 Among these, SPME combined with gas chromatography-mass spectrometry (GCMS) is the most widely used method. This approach is frequently employed to analyze volatile compounds in edible insects and their extracts (Table 5).
SPME is a clean and safe sorption technique because it requires no solvents.70 It is a cost-effective approach that allows for easy automation and enhanced repeatability.71 Hence, SPME is one of the most widely used techniques for analyzing volatiles in food. This process involves inserting and exposing the SPME fiber into the headspace, allowing the adsorption of volatile compounds. The analytes were then thermally desorbed at the GC injector port. A more recent method, known as SPME-Arrow, uses an outer tube containing a steel stick with an arrow-shaped tip. The larger diameter and extended length of the sorbent phase and fibers enhance both sensitivity and robustness.12,67,70 Choosing a suitable fiber coating is crucial for SPME and SPME-Arrow analyses. The suitability of a fiber coating for an analyte depends on the properties of the extraction phase, which affect the selectivity and reliability of the technique.73 Divinylbenzene-carboxenpolydimethylsiloxane (DVB/CAR/PDMS) is the most commonly used fiber,2,11,67,72,73 and GC is the most widely employed technique for analyzing pre-extracted volatile compounds. While nitrogen, helium, and hydrogen are commonly employed as carrier gases, helium is the exclusive choice for edible insect volatile analysis (Table 5).
Volatile compounds are generated through various intricate reactions, including carbohydrate fermentation and proteolytic and lipolytic processes. In addition, peptides, amino acids, and fatty acids are the key substrates for subsequent reactions, such as oxidation and Strecker degradation.1 Many volatile compounds have been identified in edible insects. These compounds include acids, alcohols, aldehydes, esters, hydrocarbons, indoles, ketones, and pyrazines. Only the compounds reported in at least three publications are presented in Table 6.
Seven aldehydes have been identified in edible insects. Aldehydes (a major class of volatile compounds in edible insects) have a significant influence on the volatile profile, particularly in imparting fatty notes.2 Aldehydes are categorized into linear, branched, and aromatic types based on their structural characteristics. Linear aldehydes are primarily derived from the oxidative degradation of unsaturated fatty acids such as oleic and linoleic acids, which are abundant in various foods.74 In particular, nearly half of the aldehydes detected in edible insects belong to this category, with heptanal, hexanal, and nonanal being the most frequently reported.2,11,20,73,75 In contrast, branched aldehydes are predominantly formed through Strecker degradation and the Maillard reaction.74 2- methylbutanal and 3-methylbutanal are the most commonly identified branched aldehydes.2,20,73,75 Aromatic aldehydes include benzaldehyde and phenylacetaldehyde. These compounds have been identified as key volatile compounds in roasted sesame oil, present in addition to the dominant pyrazines.76 In addition, phenylacetaldehyde (recognized as a precursor to floral notes) is also associated with the Maillard reaction in nuts and has been identified in freshly roasted almonds.77
Six hydrocarbons have been identified. However, due to their relatively high detection thresholds, these compounds are considered to have a minimal impact on the overall volatile profile of edible insects.1 Hydrocarbons such as d-limonene, o-xylene, and pxylene have been frequently detected in edible insects and edible insects-derived oils.2,11,20,48,73,75 Five acids have been reported in at least three studies: 3- methylbutanoic acid, acetic acid, butanoic acid, hexanoic acid, and propanoic acid. Acetic acid was identified in most edible insects.2,48,73,75 In addition, 2,5-dimethylpyrazine, a compound of the pyrazine group, was detected in various edible insects.2,11,48,75 This compound has also been identified in several foods, such as peanut oil and roasted sesame oil,76,78 indicating a strong association with roasted and nutty notes.76
Consumers still exhibit an aversion to edible insects, possibly due to hygienic concerns and cultural practices.79 Increasing awareness of appealing volatile compounds, superior nutritional value, and enhanced functionality can improve consumer acceptance of edible insect-based foods.13 Edible insects exhibit undesirable volatile characteristics, including acidic, cheesy, and rancid notes.2,48,73,75 Therefore, while processing the edible insects, implementing strategies to effectively control the undesirable volatile characteristics is necessary. Fermentation is recognized as an effective processing method that improves the nutritional, functional, and sensory properties of food products.80 Fermentation is also utilized to enhance the volatile profiles of edible insects, as it modifies and improves volatile compounds.
Solid-phase fermentation of defatted P. brevitarsis larval powder using lactic acid bacteria and yeast was investigated. As a result of fermentation with Lactococcus lactis, tetramethylpyrazine (nutty, vanilla, and cocoa) and trimethylpyrazine (cocoa and potato) were formed. In addition, ethyl isovalerate (apple, citrus, and pineapple), isoamyl butyrate (fruity, apricot, and banana), and 2-isobutyl-3-methylpyrazine (herbal, green, and sugar) were detected in the yeast (Saccharomyces cerevisiae) fermentation group. These compounds positively influence consumer preferences and have been shown to enhance the volatile profiles of edible insects through fermentation.13
Solid-phase fermentation of A. dichotoma larval powder using LAB was examined. Before fermentation, indole, 1-octen-3-ol, and 3-octanol were detected in A. dichotoma larvae.12 These are undesirable volatile compounds that exhibit unpleasant characteristics, such as fecal and animal notes, which negatively affect wine quality.81,82 These levels decreased following the fermentation of A. dichotoma larvae with Lactobacillus acidophilus and Lactobacillus plantarum. In addition, significant amounts of 1-propanol, 2- propanol, and ethanol were detected. These compounds contribute to the sweetness during fermentation.15
The effects of the spontaneous and sourdough fermentation of A. domesticus powder using L. plantarum have also been reported. Spontaneous fermentation resulted in a higher production of benzoic acid (faint and balsam) and disulfide dimethyl (garlic). Sourdough fermentation produces 3-methyl-1-butanol (whiskey, malt, and burnt), 2-methyl-5-propan-2- ylcyclohex-2-en-1-one (minty, caraway, and bread), and benzaldehyde (almonds and fruits) in addition. These compounds are found in alcoholic beverages, such as baijiu, and contribute to improving consumer acceptance.83,84 This suggests that fermentation is an effective method for enhancing the volatile profiles of edible insects.28
With the growing global demand for sustainable alternative foods, edible insect-derived ingredients are increasingly being integrated into a variety of processed food products, including snacks, baked goods, and meat analogs. These applications not only aim to enhance nutritional profiles but also to improve environmental sustainability. However, enhancing consumer perception remains a long-term challenge for the edible insect-based food industry. While the consumption of whole, processed edible insects has been prevalent in traditional practices, contemporary consumers demonstrate a stronger preference for edible insect-derived ingredients in powdered or visually unrecognizable forms.
Appropriate processing technologies, such as the formulation of composite fortified foods with edible insect powder and other ingredients, facilitate the effective integration of edible insects into conventional food products. Several studies have demonstrated the potential of edible insect-based ingredients as viable alternatives to traditional protein sources. For example, burgers made with mealworm-based patties received higher ratings in taste and overall acceptability than fully plant-based alternatives, suggesting positive consumer receptivity toward edible insect-derived meat substitutes.85 Similarly, the incorporation of yellow mealworm powder into maize tortillas significantly enhanced the protein content of the final product.86 Recent advances have even demonstrated the feasibility of using 4D printing technology to create edible snacks enriched with cricket powder.87 Furthermore, supplementing cookies with edible insect powders— such as T. molitor larvae, P. brevitarsis larvae, and G. bimaculatus—has been shown to enhance crude protein content while maintaining favorable sensory qualities.88 The inclusion of silkworm pupae in Frankfurt sausage formulations has likewise resulted in improved protein levels and enhanced textural properties compared to control samples.89 These diverse applications underscore the versatility of edible insects as food ingredients, facilitating their broader acceptance and integration into food systems.
Edible insects are being increasingly recognized as a sustainable and nutrient-rich alternative protein source, with significant potential in the food industry. This review examines the techniques for extracting proteins and oils from edible insects, emphasizing their efficiency and functionality. Various protein extraction methods, including pH-based solubilization, solvent-based extraction, and ultrasound-assisted techniques, have been evaluated for their effects on protein recovery. Defatting, which is commonly performed using hexane, significantly enhanced protein yield. Additionally, extraction processes involving the use of ascorbic acid and saline solutions have demonstrated improved extraction efficiencies without requiring pH adjustments. SFE, PLE, and UAE have been identified as eco-friendly and efficient oil extraction techniques. The compositional characteristics of edible insect-derived peptides and amino acids were also discussed. In particular, bioactive peptides with ACE inhibitory activity were identified in T. molitor and P. brevitarsis larvae, indicating their potential as functional food ingredients. Furthermore, the fatty acid composition of edible insects, particularly the presence of essential fatty acids such as linoleic acid and α-linolenic acid, emphasizes their nutritional value. The polyunsaturated to PUFA/SFA ratio in edible insect oils exceeded the recommended dietary threshold, reinforcing their role in promoting cardiovascular health. The characterization of edible insect volatiles revealed the role of various compounds in influencing the volatile profiles. Techniques such as SPME-GC-MS, particularly with the improved sensitivity of SPME-Arrow, have been widely employed for volatile profiling. Key volatile compounds, including aldehydes, hydrocarbons, acids, and pyrazines, were identified. In particular, fermentation modifies the volatile profiles of edible insects by generating desirable volatile compounds, suggesting its potential to enhance the sensory quality of edible insect-based food products.
C. Perez-Santaescolastica, A. De Winne, J. Devaere, and I. Fraeye, Trends Food Sci. Technol., 127, 352-367 (2022). https://doi.org/10.1016/j.tifs.2022.07.011 https://doi.org/10.1016/j.tifs.2022.07.011
J. K. Nam, B. M. Kwak, and H. W. Jang, Food Chem., 474, 143237 (2025). https://doi.org/10.1016/j.foodchem. 2025.143237 https://doi.org/10.1016/j.foodchem.2025.143237
V. A. Cruz, C. M. Vicentini-Polette, D. R. Magalhaes, and A. L. de Oliveira, Food Chem., 463, 141199 (2025). https://doi.org/10.1016/j.foodchem.2024.141199 https://doi.org/10.1016/j.foodchem.2024.141199
A. van Huis, J. van Itterbeeck, H. Klunder, E. Mertens, A. Halloran, G. Muir, and P. Vantomme, 'Edible insects: Future prospects for food and feed security', Food and Agriculture Organization of the United Nations, Rome (2013). https://www.cabidigitallibrary.org/doi/full/10.5555/201332170745 https://doi.org/10.5555/201332170745
A. J. da Silva Lucas, L. M. de Oliveira, M. Da Rocha, and C. Prentice, Food Chem., 311, 126022 (2020). https://doi.org/10.1016/j.foodchem.2019.126022 https://doi.org/10.1016/j.foodchem.2019.126022
A. Baiano, Trends Food Sci. Technol., 100, 35-50 (2020). https://doi.org/10.1016/j.tifs.2020.03.040 https://doi.org/10.1016/j.tifs.2020.03.040
N. Meijer, R. A. Safitri, W. Tao, and E. F. Hoek-Van den Hil, Anim., 19, 101468 (2025). https://doi.org/10.1016/j.animal.2025.101468 https://doi.org/10.1016/j.animal.2025.101468
D. Raheem, C. Carrascosa, O. B. Oluwole, M. Nieuwland, A. Saraiva, R. Millán, and A. Raposo, Crit. Rev. Food Sci. Nutr., 59(14), 2169-2188 (2019). https://doi.org/10.1080/10408398.2018.1440191 https://doi.org/10.1080/10408398.2018.1440191
Z. Q. Zhang, S. C. Chen, J. H. Xiao, and D. W. Huang, Food Biosci., 59, 103879 (2024). https://doi.org/10.1016/j.fbio.2024.103879 https://doi.org/10.1016/j.fbio.2024.103879
J. H. Lee, T. K. Kim, C. H. Jeong, H. I. Yong, J. Y. Cha, B. K. Kim, and Y. S. Choi, Food Sci. Biotechnol., 30,1003-1023 (2021). https://doi.org/10.1007/s10068-021-00942-8 https://doi.org/10.1007/s10068-021-00942-8
M. H. Lee, T. K. Kim, J. Y. Cha, H. I. Yong, M. C. Kang, H. W. Jang, and Y. S. Choi, LWT, 167, 113888 (2022). https://doi.org/10.1016/j.lwt.2022.113888 https://doi.org/10.1016/j.lwt.2022.113888
J. Y. Lee, W. S. Kim, Y. Y. Lee, Y. S. Choi, H. Choi, and H. W. Jang, J. Sep. Sci., 42(18), 2942-2948 (2019). https://doi.org/10.1002/jssc.201900388 https://doi.org/10.1002/jssc.201900388
J.Y.Cha, J.Han,J.Heo, H.H. Yu, Y.J.Kim, H. W.Jang, M. R. Kim, and Y. S. Choi, Food Chem., 452, 139480 (2024). https://doi.org/10.1시10chem.2024.139480 https://doi.org/10.1시10chem.2024.139480
D. L. García-González, N. Tena, R. Aparicio-Ruiz, and M. T. Morales, Meat Sci., 80(2), 315-325 (2008). https://doi.org/10.1016/j.meatsci.2007.12.015 https://doi.org/10.1016/j.meatsci.2007.12.015
H. E. Lee, J. Kim, Y. Kim, W. Y. Bang, J. Yang, S. J. Lee, and Y. H. Jung, Food Biosci., 43, 101257 (2021). https://doi.org/10.1016/j.fbio.2021.101257 https://doi.org/10.1016/j.fbio.2021.101257
T. K. Kim, H. I. Yong, C. H. Jeong, S. G. Han, Y. B. Kim, H. D. Paik, and Y. S. Choi, Food Sci. Anim. Res., 39(4), 643 (2019). https://doi.org/10.5851/kosfa.2019.e56 https://doi.org/10.5851/kosfa.2019.e56
B. Carriço-S, C. S. Teixeira, C. Villa, E. Mendes, I. M. Ferreira, I. Mafra, and J. Costa, Food Chem., 468, 142453 (2025). https://doi.org/10.1016/j.foodchem.2024.142453 https://doi.org/10.1016/j.foodchem.2024.142453
J. H. Lee, Y. J. Kim, T. K. Kim, K. M. Song, and Y. S. Choi, Food Chem., 437, 137852 (2024). https://doi.org/10.1016/j.foodchem.2023.1378521 https://doi.org/10.1016/j.foodchem.2023.1378521
M. Mishyna, J. J. I. Martinez, J. Chen, and O. Benjamin, Food Res. Int., 116, 697-706 (2019). https://doi.org/10.1016/j.foodres.2018.08.098 https://doi.org/10.1016/j.foodres.2018.08.098
J. H. Lee, J. Y. Cha, T. K. Kim, Y. S. Choi, and H. W. Jang, LWT, 151, 112095 (2021). https://doi.org/10.1016/j.lwt.2021.112095 https://doi.org/10.1016/j.lwt.2021.112095
B. D. Choi, N. A. Wong, and J. H. Auh, Korean J. Food Sci. Anim. Resour., 37(6), 955 (2017). https://doi.org/10.5851/kosfa.2017.37.6.955 https://doi.org/10.5851/kosfa.2017.37.6.955
T. K. Kim, H. I. Yong, Y. B. Kim, S. Jung, H. W. Kim, and Y. S. Choi, Food Chem., 336, 127679 (2021). https://doi.org/10.1016/j.foodchem.2020.127679 https://doi.org/10.1016/j.foodchem.2020.127679
Y. Bai, Q. Zhao, M. He, X. Ye, and X. Zhang, J. Pharm. Biomed. Anal., 163, 78-87 (2019). https://doi.org/10.1016/j.jpba.2018.09.033 https://doi.org/10.1016/j.jpba.2018.09.033
M. Montowska, P. Ł. Kowalczewski, I. Rybicka, and E. Fornal, Food Chem., 289, 130-138 (2019). https://doi.org/10.1016/j.foodchem.2019.03.062 https://doi.org/10.1016/j.foodchem.2019.03.062
C. Dai, H. Ma, L. Luo, and X. Yin, Eur. Food Res. Technol., 236, 681-689 (2013). https://doi.org/10.1007/s00217-013-1923-z https://doi.org/10.1007/s00217-013-1923-z
A. Brai, C. I. Trivisani, C. Vagaggini, R. Stella, R. Angeletti, G. Iovenitti, V. Francardi, and E. Dreassi, Food Chem., 393, 133409 (2022). https://doi.org/10.1016/j.foodchem.2022.133409 https://doi.org/10.1016/j.foodchem.2022.133409
J. H. Lee, T. K. Kim, H. I. Yong, J. Y. Cha, K. M. Song, H. G. Lee, J. G. Je, M. C. Kang, and Y. S. Choi, Food Chem., 399, 133897 (2023). https://doi.org/10.1016/j.foodchem.2022.133897 https://doi.org/10.1016/j.foodchem.2022.133897
B. T. B. Vasilica, M. S. Chiș, E. Alexa, C. Pop, A. Păucean, S. Man, M. Igual, K. M. Haydee, K. E. Dalma, S. Sanila, S. Socaci, A. Faracas, A. Berbecea, I. Popescu, and S. Muste, Insects, 13(7), 576 (2022). https://doi.org/10.3390/insects13070576 https://doi.org/10.3390/insects13070576
S. Kittibunchakul, K. Whanmek, and C. Santivarangkna, LWT, 189, 115444 (2023). https://doi.org/10.1016/j.lwt. 2023.115444 https://doi.org/10.1016/j.lwt.2023.115444
B. K. Mintah, R. He, A. A. Agyekum, M. Dabbour, M. K. Golly, and H. Ma, Food Process Eng., 43(4), e13362 (2020). https://doi.org/10.1111/jfpe.13362 https://doi.org/10.1111/jfpe.13362
D. Zhu, X. Huang, F. Tu, C. Wang, and F. Yang, J. Food Biochem., 44(5), e13186 (2020). https://doi.org/10.1111/jfbc.13186 https://doi.org/10.1111/jfbc.13186
T. K. Kim, H. I. Yong, H. H. Chun, M. A. Lee, Y. B. Kim, and Y. S. Choi, J. Asia Pac. Entomol., 23(2), 298-305 (2020). https://doi.org/10.1016/j.aspen.2019.12.017 https://doi.org/10.1016/j.aspen.2019.12.017
H. Tomotake, M. Katagiri, and M. Yamato, J. Nutr Sci. Vitaminol., 56(6), 446-448 (2010). https://doi.org/ 10.3177/jnsv.56.446 https://doi.org/10.3177/jnsv.56.446
M. D. Finke, Zoo Biol., 26(2), 105-115 (2007). https://doi.org/10.1002/zoo.20123 https://doi.org/10.1002/zoo.20123
G. Wu, H. R. Cross, K. B. Gehring, J. W. Savell, A. N. Arnold, and S. H. McNeill, J. Anim. Sci., 94(6), 2603-2613 (2016). https://doi.org/10.2527/jas.2016-0478 https://doi.org/10.2527/jas.2016-0478
F. Zhao, P. Zhuang, C. Song, Z. H. Shi, and L. Z. Zhang, Food Chem., 118(2), 224-227 (2010). https://doi.org/10.1016/j.foodchem.2009.04.110 https://doi.org/10.1016/j.foodchem.2009.04.110
M. E. Parker, S. Zobrist, H. E. Lutterodt, C. R. Asiedu, C. Donahue, C. Edick, K. Mansen, G. Pelto, P. Milani, S. Soor, A. Laar, and C. M. Engmann, BMC Nutr., 6, 1-11 (2020). https://doi.org/10.1186/s40795-020-0331-6 https://doi.org/10.1186/s40795-020-0331-6
C. J. Zhao, A. Schieber, and M. G. Gänzle, Food Res. Int.,89, 39-47 (2016). https://doi.org/10.1016/j.foodres.2016.08.042 https://doi.org/10.1016/j.foodres.2016.08.042
J. L. Martina, J. M. Martinez, and J. A. Olarte, Appetite, 55(1), 181-187 (2010). https://doi.org/10.1016/j.appet.2010.05.002 https://doi.org/10.1016/j.appet.2010.05.002
W. T. Chitisankul, M. Murakami, C. Tsukamoto, and K. Shimada, LWT, 115, 108432 (2019). https://doi.org/10.1016/j.lwt.2019.108432 https://doi.org/10.1016/j.lwt.2019.108432
Y. W. Li and B. Li, J. Theor. Biol., 318, 29-43 (2013). https://doi. org/10.1016/j.jtbi.2012.10.029 https://doi.org/10.1016/j.jtbi.2012.10.029
Y. Y. Li, Y. Z. Fan, J. L. Liu, Z. S. Meng, A. X. Huang, F. R. Xu, and X. Wang, Food Res. Int, 164, 112382 (2023). https://doi.org/10.1016/j.foodres.2022.112382 https://doi.org/10.1016/j.foodres.2022.112382
Y. H. Lin, J. S. Tsai, and G. W. Chen, J. Food Biochem., 41(4), e12385 (2017). https://doi.org/10.1111/jfbc.12385 https://doi.org/10.1111/jfbc.12385
M. Alfosea-Simón, S. Simón-Grao, E. A. Zavala-Gonzalez, J. M. Cámara-Zapata, I. Simón, J. J. Martínez-Nicolás, V. Marina, and F. García-Sánchez, Front. Plant Sci., 11,581234 (2021). https://doi.org/10.3389/fpls.2020.581234 https://doi.org/10.3389/fpls.2020.581234
Y. Hou and G. Wu, Adv. Nutr, 9(6), 849-851 (2018). https://doi.org/10.1093/advances/nmy054 https://doi.org/10.1093/advances/nmy054
X. Zhuang, Z. Zhang, Y. Wang, and Y. Li, Ind. Crops Prod., 126, 340-346 (2018). https://doi.org/10.1016/j.indcrop.2018.10.004 https://doi.org/10.1016/j.indcrop.2018.10.004
G. E. Lee, Y. J. Kim, H. B. Jang, J. Y. Cha, Y. S. Choi, and H. W. Jang, J. Insects Food Feed, 9, 1631-1642 (2023). https://doi.org/10.1163/23524588-20230039 https://doi.org/10.1163/23524588-20230039
P. Otero, A. Gutierrez-Docio, J. N. Del Hierro, G. Reglero, and D. Martin, Food Chem., 314, 126200 (2020). https://doi.org/10.1016/j.foodchem.2020.126200 https://doi.org/10.1016/j.foodchem.2020.126200
R. Conte, L. M. Gullich, D. Bilibio, O. Zanella, J. P. Bender, N. Carniel, and W. L. Priamo, Food Chem., 213, 425-430 (2016). https://doi.org/10.1016/j.foodchem.2016.06.111 https://doi.org/10.1016/j.foodchem.2016.06.111
J. N. Del Hierro, A. Gutiérrez-Docio, P. Otero, G. Reglero, and D. Martin, Food Chem., 309, 125742 (2020). https://doi.org/10.1016/j.foodchem.2019.125742 https://doi.org/10.1016/j.foodchem.2019.125742
K. Kumar, S. Srivastav, and V. S. Sharanagat, Ultrason. Sonochem., 70, 105325 (2021). https://doi.org/10.1016/j.ultsonch.2020.105325 https://doi.org/10.1016/j.ultsonch.2020.105325
C. Perez-Santaescolastica, I. de Pril, I. van de Voorde, and I. Fraeye, Foods, 12(22), 4090 (2023). https://doi.org/10.3390/foods12224090 https://doi.org/10.3390/foods12224090
A. Orkusz, L. Dymińska, K. Banaś, and J. Harasym, Foods, 13(1), 32 (2023). https://doi.org/10.3390/foods13010032 https://doi.org/10.3390/foods13010032
A. Osimani, C. Garofalo, V. Milanović, M. Taccari, F. Cardinali, L. Aquilanti, M. Pasquini, M. Mozzon, N. Raffaelli, S. Ruschioni, P. Riolo, N. Isidoro, and F. Clementi, Eur. Food Res. Technol., 243, 1157-1171 (2017). https://doi.org/10.1007/s00217-016-2828-4 https://doi.org/10.1007/s00217-016-2828-4
K. M. C. Nogoy, H. J. Kim, Y. Lee, Y. Zhang, J. Yu, D. H. Lee, X. Z. Li, S. B. Smith, H. A. Seong, and S. H. Choi, Food Sci. Nutr., 8(7), 3617-3625 (2020). https://doi.org/10.1002/fsn3.1644 https://doi.org/10.1002/fsn3.1644
C. Dorni, P. Sharma, G. Saikia, and T. Longvah, Food Chem., 238, 9-15 (2018). https://doi.org/10.1016/j.foodchem.2017.05.072 https://doi.org/10.1016/j.foodchem.2017.05.072
V. Ivanova-Petropulos, S. Mitrev, T. Stafilov, N. Markova, E. Leitner, E. Lankmayr, and B. Siegmund, Food Res. Int., 77, 506-514 (2015). https://doi.org/10.1016/j.foodres.2015.08.014 https://doi.org/10.1016/j.foodres.2015.08.014
M. Geranpour, E. Assadpour, and S. M. Jafari, Trends Food Sci. Technol., 102, 71-90 (2020). https://doi.org/10.1016/j.tifs.2020.05.028 https://doi.org/10.1016/j.tifs.2020.05.028
S. Rajaram, Am. J. Clin. Nutr., 100, 443S-448S (2014). https://doi.org/10.3945/ajcn.113.071514 https://doi.org/10.3945/ajcn.113.071514
Q. Yuan, F. Xie, W. Huang, M. Hu, Q. Yan, Z. Chen, Y. Zheng, and L. Liu, Phytother. Res., 36(1), 164-188 (2022). https://doi.org/10.1002/ptr.7295 https://doi.org/10.1002/ptr.7295
Z. Zeng, X. Qin, H. Wang, Z. Chen, D. Huang, D. Xiang, and X. Liu, LWT, 192, 115736 (2024). https://doi.org/10.1016/j.lwt.2024.115736 https://doi.org/10.1016/j.lwt.2024.115736
L. Zhang, K. Zhang, H. Yang, K. Yue, R. Liu, Y. Bi, and C. Ma, J. Food Compos. Anal., 115, 105021 (2023). https://doi.org/10.1016/j.jfca.2022.105021 https://doi.org/10.1016/j.jfca.2022.105021
J. B. German, Mater. Child Nutr., 7, 2-16 (2011). https://doi.org/10.1111/j.1740-8709.2011.00300.x https://doi.org/10.1111/j.1740-8709.2011.00300.x
Z. J. Wang, G. M. Li, B. M. Nie, Y. Lu, and M. Yin, Chem. Biol. Interact., 160(1), 80-87 (2006). https://doi.org/10.1016/j.cbi.2005.12.0086 https://doi.org/10.1016/j.cbi.2005.12.0086
B. Karsli, J. Food Compost. Anal., 103, 104105 (2021). https://doi.org/10.1016/j.jfca.2021.104105 https://doi.org/10.1016/j.jfca.2021.104105
67 N. E. Song, J. Y. Lee, Y. Y. Lee, J. D. Park, and H. W. Jang, Appl. Biol. Chem., 62, 1-8 (2019). https://doi.org/10.1186/s13765-019-0424-6 https://doi.org/10.1186/s13765-019-0424-6
Kiatbenjakul, P., Intarapichet, K. O., and Cadwallader, K. R, Flavour Fragr. J., 29(2), 107-113 (2013). https://doi.org/10.1002/ffj.3185 https://doi.org/10.1002/ffj.3185
Kiatbenjakul, P., Intarapichet, K. O., and Cadwallader, K. R, Food Chem., 168, 639-647 (2015). https://doi.org/10.1016/j.foodchem.2014.07.108 https://doi.org/10.1016/j.foodchem.2014.07.108
J. Y. Cha, Y. W. Chin, J. Y. Lee, T. W. Kim, and H. W. Jang, Foods, 9(10), 1422 (2020). https://doi.org/10.3390/foods9101422 https://doi.org/10.3390/foods9101422
J. K. Nam, J. Y. Lee, and H. W. Jang, Food Chem. X, 23, 101576 (2024). https://doi.org/10.1016/j.fochx.2024.101576 https://doi.org/10.1016/j.fochx.2024.101576
A. R. Mansur, H. J. Lee, H. K. Choi, T. G. Lim, M. Y. Yoo, H. W. Jang, and T. G. Nam, J. Food Process. Preserv., 42(10), e13746 (2018). https://doi.org/10.1111/jfpp.137467 https://doi.org/10.1111/jfpp.137467
J. Y. Cha, T. K. Kim, Y. J. Kim, J. H. Lee, M. C. Kang, H. W. Jang, and Y. S. Choi, Future Foods, 10, 100429 (2024). https://doi.org/10.1016/j.fufo.2024.100429 https://doi.org/10.1016/j.fufo.2024.100429
F.B. Whitfield and D. S. Mottram, Crit. Rev. FoodSci. Nutr., 31(1-2), 1-58 (1992). https://doi.org/l0.1080/10408399209527560 https://doi.org/1080/10408399209527560
C. Perez-Santaescolastica, A. De Winne, J. Devaere, and I. Fraeye, Food Res. Int., 164, 112389 (2023). https://doi.org/10.1016/j.foodres.2022.112389 https://doi.org/10.1016/j.foodres.2022.112389
W. T. Yin, X. T. Ma, S. J. Li, X. D. Wang, H. M. Liu, and R. Shi, Food Res. Int., 150, 110794 (2021). https://doi.org/10.1016/j.foodres.2021.110794 https://doi.org/10.1016/j.foodres.2021.110794
A. Valdés García, R. Sánchez Romero, A. Juan Polo, S. Prats Moya, S. E. Maestre Pérez, and A. Beltrán Sanahuja, Foods, 10, 1611 (2021). https://doi.org/10.3390/foods10071611 https://doi.org/10.3390/foods10071611
Q.Dun,L. Yao,Z.Deng,H.Li,J.Li,Y.Fan, andB.Zhang, LWT, 112, 107648 (2019). https://doi.org/l0.1016/j.lwt.2018.11,084 https://doi.org/1016/j.lwt.2018.11
W. Verbeke, Food Qual. Prefer., 39, 147-155 (2015). https://doi.org/10.1016/j.foodqual.2014.07.008 https://doi.org/10.1016/j.foodqual.2014.07.008
A. K. Nedele, S. Gross, M. Rigling, and Y. Zhang, Food Chem., 334, 127591 (2021). https://doi.org/10.1016/j.foodchem.2020.127591 https://doi.org/10.1016/j.foodchem.2020.127591
J. Gong, Q. Zuo, Z. Wu, C.Zhao,J. Wei, and Y.Huang, Food Chem. X, 23, 101660 (2024). https://doi.org10.1016/jfochx.2024,101660 https://doi.org/10.1016/jfochx.2024
L. Delcros, A. Costis, C. Le Guerneve, S. Collas, M. Herv, and A. Roland, Food Chem., 413, 135678 (2023). https://doi.org/10.1016/j.foodchem.2023.135678 https://doi.org/10.1016/j.foodchem.2023.135678
H. Huang, Y. Wu, H. Chen, Y. Hou, J. Wang, J. Hong, and B. Sun, J. Sci. Food Agric., 103(15), 7434–7444 (2023). https://doi.org/10.1002/jsfa.12823 https://doi.org/10.1002/jsfa.12823
S. Liang, Y. Liu, S. Yuan, Y. Liu, B. Zhu, and M. Zhang, Foods, 11(15), 2224 (2022). https://doi.org/10.3390/foods11152224 https://doi.org/10.3390/foods11152224
R. C. Megido, C. Gierts, C. Blecker, Y. Brostaux, É. Haubruge, T. Alabi, and F. Francis, Food Qual. Prefer., 52, 237-243 (2016). https://doi.org/10.1016/j.foodqual.2016.05.004 https://doi.org/10.1016/j.foodqual.2016.05.004
E. D. Aguilar-Miranda, M. G. López, C. Escamilla-Santana, and A. P. Barba de la Rosa, J. Agric. Food Chem., 50(1), 192-195 (2002). https://doi.org/10.1021/jf010691y8 https://doi.org/10.1021/jf010691y8
Z. Kang, Z. Wang, W. Zhang, and Z. Fang, Food Biosci., 55, 102971 (2023). https://doi.org/10.1016/j.fbio.2023.102971 https://doi.org/10.1016/j.fbio.2023.102971
H. B. Jang, J. Y. Baek, Y. S. Choi, and H. W. Jang, Korean J. Food Sci. Technol., 54(2), 171-178 (2022). https://doi.org/10.9721/KJFST.2022.54.2.171 https://doi.org/10.9721/KJFST.2022.54.2.171
Y. S. Park, Y. S. Choi, K. E. Hwang, T.K. Kim, C.W. Lee, D. M. Shin, and S. G. Han, Korean J. Food Sci. Anim. Resour., 37(3), 351-359 (2017). https://doi.org10.5851/kosfa.2017.37.3.351 https://doi.org/10.5851/kosfa.2017.37.3.351