
- P-ISSN 1225-0163
- E-ISSN 2288-8985



Capillary electrophoresis–inductively coupled plasma mass spectrometry (CE-ICP-MS) is a highly effective technique for elemental and chemical speciation analysis. However, coupling CE with ICP-MS presents significant challenges due to maintaining electrical contact at the CE outlet while ensuring efficient transfer of the electrophoretically separated analytes into the mass spectrometer. This interface design is particularly complex, as it must support the formation of an electric field intrinsic to CE while being compatible with the sample introduction system of ICP-MS. Over the past three decades, extensive research efforts have led to the development of various interface configurations, resulting in numerous successful applications and scientific advancements. This article provides an overview of CE-ICP-MS to support the broader adoption of the technique. It introduces the fundamental principles of capillary electrophoresis and outlines the key components of the ICP-MS instrumentation. It also examines the primary interface strategies that have been developed to effectively couple CE with ICP-MS. In addition, recent research trends over the past decade are reviewed to highlight the expanding scope and potential of CE-ICP-MS in modern analytical science.
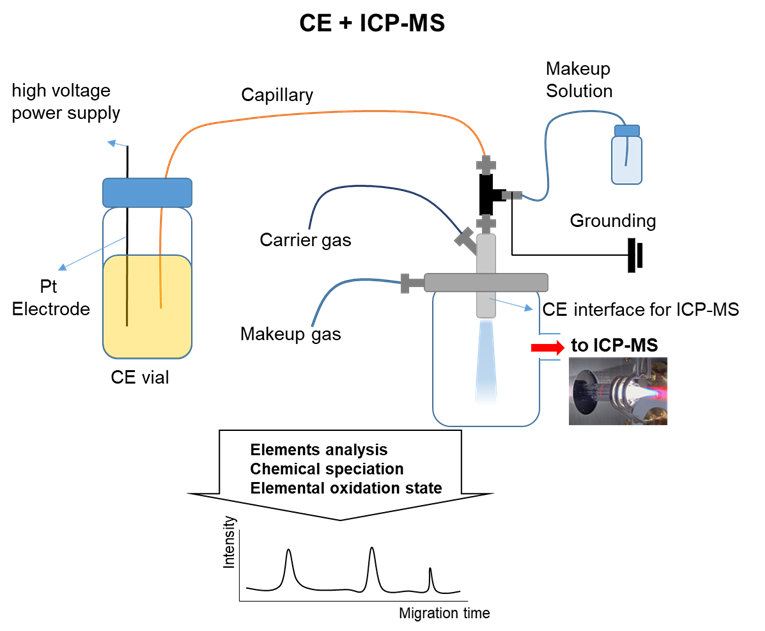
Inorganic analysis for the determination of elemental composition plays a critical role in scientific and industrial development, as well as in radioisotope research. It serves both qualitative identification and quantitative measurement purposes. However, as elements may exist in diverse chemical forms—such as inorganic salts, organometallic compounds, and metal complexes—quantifying only their total concentration is often inadequate for a comprehensive understanding of their chemical behavior or biological function. Therefore, elucidating the oxidation state and chemical speciation of elements is of critical mportance. Elemental speciation analysis encompasses the separation, identification, and quantification of the distinct chemical forms of an element within a given sample. Such analyses yield essential insights into elemental toxicity, bioavailability, and metabolic pathways.1-3 Various analytical methods are available for element separation and detection. Ion chromatography has traditionally been the method of choice,4 but capillary electrophoresis (CE) has emerged as a powerful alternative.5 In contrast to conventional column-based techniques, CE utilizes open tubular capillaries, eliminating the need for a stationary phase and enabling rapid, versatile analysis with a single capillary. Additionally, CE offers benefits such as easier maintenance, straightforward sample handling, and significantly reduced chemical consumption and waste. Following separation, detection is commonly performed using UV-Vis spectroscopy. However, due to the lack of chromophores in many inorganic species, more sensitive and selective methods like inductively coupled plasma mass spectrometry (ICPMS) are often preferred. ICP-MS offers exceptional sensitivity, a wide dynamic range, the ability to detect multiple elements simultaneously, and precise isotopic analysis. When CE is coupled with ICP-MS, the resulting hybrid system leverages the advantages of both methods, providing a robust solution for detailed elemental speciation.1-3,6 This study presents a comprehensive overview of the underlying principles of CE and ICP-MS, examines critical factors involved in their successful integration, and elucidates applications demonstrating the analytical potential of the combined technique.
CE is widely used as a high-performance separation technique for a variety of substances, ranging from small molecules such as metal ions, organic ions, and drugs to large biomolecules such as proteins and DNA. This is because it offers several advantages, such as high separation efficiency, rapid analysis, low sample consumption, and easy automation. Since the introduction of modern capillary electrophoresis by Jorgenson and Lukas in 1981, the initial decade focused on developing commercially available instruments and establishing it as a robust analytical technique.7 In the 1990s, it was utilized for rapid, high-resolution DNA analysis, playing a key role in the early achievement of the Human Genome Project's research objectives. CE is currently applied in a wide range of analyses, from small inorganic ions to large biomolecules. Today, tens of thousands of CE instruments are in use globally, and a search for “capillary electrophoresis” on the Web of Science from 2021 to 2024 yielded over 4,600 published studies. As its applications continue to expand, CE is solidifying its position as a standard analytical technique.
In CE, electrophoresis describes the movement of charged molecules within a solution under the influence of an electric field. The speed and direction of movement are influenced by the size and charge of the ion, as well as the charge polarity.8 Additionally, the electroosmotic flow (EOF), which is the flow of the electrophoresis buffer through the capillary due to the electric field, also affects the separation.8 When the capillary inner wall surface is ionized or buffer ions adsorb onto the capillary inner wall, the surface of the capillary becomes charged, attracting ions in the buffer solution to the inner surface. This creates an electrical double layer, consisting of a fixed layer tightly bound to the capillary wall and a more mobile diffuse layer. EOF occurs when the diffuse layer is drawn towards the electrode upon application of an electric field, causing the entire buffer solution to move towards the electrode. Generally, EOF is directed towards the cathode, and solutes are also affected by this EOF. Cations move faster than the EOF because their electrophoretic movement is in the same direction, while neutral molecules have no electrophoretic mobility and move at the same rate as the EOF. Anions can also migrate toward the cathode if the electroosmotic mobility of the buffer exceeds the anion’s electrophoretic mobility. Consequently, CE enables the simultaneous separation of both cations and anions in a single run. The elution order from the capillary is cations, neutral molecules, and anions, with each component separated according to its chargeto-size ratio.
The electrophoretic velocity (vEP) is directly proportional to the electric field (E), as described by the following equation:
where vEP represents electrophoretic velocity, μEP is the electrophoretic mobility, and E notes the applied electric field. Electrophoretic mobility is defined by the following equation, yielding a unique value based on the ratio of an ion's charge to its size:
where μEP represents the electrophoretic mobility, q is the ionic charge, η is the buffer viscosity, and γ denotes the ionic radius. By combining this fundamental principle of electrophoresis with various techniques, different modes of electrophoresis have been developed and widely applied. Commonly utilized modes include capillary zone electrophoresis (CZE), micellar electrokinetic capillary chromatography (MEKC), capillary gel electrophoresis (CGE), capillary isoelectric focusing (CIEF), and capillary isotachophoresis (CITP). The diversity of these modes has positioned electrophoresis as a versatile and powerful separation and analytical method. The basic electrophoretic apparatus is relatively straightforward as shown in Fig. 1. Its main features include a sample vial, an inlet vial (source vial), an outlet vial (destination vial), a capillary, a detector, a high-voltage power supply, and a data acquisition and processing unit. Ideally, the capillary should possess chemical and physical stability, a small inner diameter, high thermal conductivity to efficiently dissipate heat (Joule heating), and transparency to ultraviolet light. The most commonly employed detection methods in electrophoresis are UV-Vis absorbance detection and laser-induced fluorescence detection. The high-voltage power supply should deliver up to 30 kV, as higher voltages enhance separation efficiency, reduce analysis time, and improve resolution. Although electrophoresis is typically performed under constant voltage, it can also be conducted under constant current, with the electric field applied by periodically switching the voltage.
The basic procedure for performing separation analysis via electrophoresis involves several key steps. First, if needed, the capillary is pretreated, the inlet and outlet vials are filled with a buffer, and the capillary is conditioned by rinsing it with the same buffer. The sample is then introduced into the capillary, followed by the application of an electric field. In CZE as a simple CE method, the sample volume introduced is typically less than 1 % of the total capillary volume. As the sample travels through the capillary, its components are separated primarily based on their charge-to-size ratios and are subsequently detected. The resulting data are sent to an output display and visualized as an electropherogram, in which the x-axis represents migration time and the yaxis corresponds to detector response (see Fig. 2). Commercial capillary electrophoresis systems usually rely on UV-Vis detector. However, a variety of alternative detection techniques have been explored in research settings. For analytes such as inorganic ions that do not absorb UV-Vis light, sensitivity can be enhanced through laser-induced fluorescence after suitable derivatization. Alternatively, inorganic ions can be directly detected using ICP-MS.
typically employs a sample introduction system designed for liquid samples. Within the inductively coupled plasma, the high-temperature plasma discharge effectively decomposes sample constituents into their elemental forms. These elements undergo ionization, forming positively charged ions that are subsequently detected by the mass spectrometer based on their mass-to-charge ratio (m/z). The sample introduction system consists of a nebulizer and a spray chamber. As the liquid sample passes through these components, it is transformed into an aerosol. passes through various heating zones within the plasma torch, where it is dried, vaporized, atomized, and ultimately ionized. The sample reaches a temperature of approximately 6,000 − 7,000 K in the analytical region of the plasma, and the excited atoms and ions are detected, allowing for the determination of the sample's elemental composition. While negative ions (e.g., halogens) can also be generated in the plasma, most commercial instruments are designed to measure positive ions due to differences in the extraction and transport processes.9 A typical ICP-MS instrument configuration is shown in Fig. 3. In the sample introduction part, a peristaltic pump delivers the liquid sample at a rate of 1 − 2 mL/min. Peristaltic pumps maintain a consistent flow rate regardless of sample viscosity. After passing through the nebulizer and spray chamber, the liquid sample is converted into a fine aerosol by a carrier gas flow (usually argon) at about 1 L/min. The spray chamber helps to reduce any pulsations caused by the peristaltic pump. Only 1 − 2 % of the liquid sample enters the plasma as an aerosol in a gaseous state; the majority of the sample remains as larger droplets, fails to vaporize, and is expelled through the bottom of the spray chamber.10 The plasma torch typically consists of three concentric quartz tubes. A gas (usually argon) flows between the outer and middle tubes at a rate of 12 − 18 L/min. An auxiliary gas, forming a second gas flow, flows between the middle tube and the sample injector at approximately 1 L/min, while a nebulizer gas, forming a third gas flow, also flows at about 1 L/ min. The ionized material then passes through the interface region, reaching the sampler cone and skimmer cone,11 before entering the mass analyzer for detection. Typically, ICP-MS is capable of achieving detection limits in the range of 0.001 to 0.05 μmol/L, depending on the on the element and analytical conditions.12 In general, quadrupole mass filter technique has been the most widely used for ion separation in mass analyzers. However, quadrupole filter technique has limitations when it comes to detecting complex matrices due to interference from other elements. Typically mass analyzers are four types: quadrupole mass filters, double-focusing magnetic sectors, time-of-flight analyzers, and instruments utilizing collision/reaction cell technique.13
Capillary electrophoresis-inductively coupled plasmamass spectrometry (CE-ICP-MS) is a relatively recent analytical technique, first developed in 1995 by the Olesik, Barnes, and Lopez-Avila research groups.14,15 One of the main challenges in designing an effective interface for this technique is the efficient transport of the small quantities of CE eluent, which are nebulized into a fine aerosol, to the plasma source. Moreover, the lack of commercially available nebulizers that function well at the low flow rates typical of CE further complicates the coupling of these two analytical instruments, and research in this field remains limited. Developing a suitable interface is essential for successful CE-ICP-MS integration. Main considerations when connecting these instruments include: maintaining effective electrical contact at the CE capillary outlet, reducing laminar flow resistance caused by the suction effect of the nebulizer,16 minimizing peak broadening, and ensuring high transport efficiency. In most instrument designs, the outlet of the CE capillary is positioned into the central tube of the nebulizer, while maintaining an electrical connection with the capillary's end. Many reported CE-ICP-MS interfaces feature a T-shaped union to balance flow rates, ensuring a sufficient flow rate to the nebulizer despite the low flow rate of the CE.2 This typically involves introducing a buffer solution to compensate for the CE flow rate (in the µL/min range), which is too low compared to the nebulizer's standard aspiration rate.1,3 The interface methods are generally categorized into two types: sheathless interfaces and sheath-flow interfaces (referred to as devices for makeup solutions).1 In addition, commercially available microflow interfaces are introduced.
The sheath-flow interface not only facilitates the efficient transport of analytes from CE to ICP-MS, but also provides a stable electrical connection between the two systems. Although the sheath-flow interface is structurally more complex than the sheathless interface, it is more commonly employed due to its reliability and practicality. The flow rate of CE is significantly lower than that of ICP-MS, which often results in suction effects at the nebulizer tip caused by the gas flow, drawing liquid from the capillary. To address this, the sheath-liquid flow interface was developed. However, this approach may lead to dilution of analytes, potentially reducing detection sensitivity. The sheath-flow interface consists of several key components. First, a separation capillary (or CE capillary) is used to transfer the CE effluent to the ICP torch. Second, the sheath liquid, which flows around the outer surface of the capillary, ensures electrical continuity. Third, a platinum ground electrode (Pt electrode) is employed to complete the CE electrical circuit. Lastly, a nebulizing gas is used to assist in the aerosolization of the liquid sample.2 Sheath-flow interfaces are typically categorized based on the arrangement of the nebulizing gas flow and the liquid flows. These are classified as either concentric nebulizers (if both flows are aligned in the same direction) or cross-flow nebulizers (if the flows are oriented perpendicularly).1 The concentric nebulizerbased interface is the most widely used configuration due to its compatibility with a wide range of experimental conditions. It can be constructed using various connectors, with the tee union configuration being one of the most representative CE–ICP-MS coupling methods. In this setup, the separation capillary outlet, the tube supplying the sheath liquid, and the tube carrying the combined flow to the nebulizer or ICP torch are joined via a tee connector. The platinum electrode is clamped to the tee using an alligator clip to maintain electrical continuity.1 The sheath liquid conducts electricity to the end of the separation capillary, thereby completing the electrical circuit and compensating for the low CE flow rate, which otherwise would be insufficient to counteract the suction generated by the nebulizer gas. This prevents the formation of laminar flow within the capillary.1, 17 In cross-nebulizer-based interfaces, similar connectors are used to integrate the separation capillary outlet, sheath liquid tubing, and platinum electrode. However, a key difference is that the outlet of the tube carrying the mixed CE effluent and sheath liquid is immersed in an organic solvent (e.g., methanol), causing it to swell and fit securely into the inlet of the cross-flow nebulizer, which is aligned perpendicularly.18 Compared to concentric nebulizers, cross nebulizers offer enhanced mechanical durability and can deliver more stable performance. Moreover, the vertically oriented gas flow in cross nebulizers continuously washes the capillary outlet, resulting in consistent and reproducible results across multiple runs.18,19
To maintain continuous electrical contact at the CE outlet without the use of a sheath liquid, sheathless interface strategies have been developed. These generally involve either coating the outer surface of the capillary with a conductive material14 or inserting a metal wire inside the capillary,20 ensuring a seamless electrical contact. In the former approach, the separation capillary is inserted through the center of a Meinhardtype nebulizer for aerosol generation. To ensure electrical continuity, approximately 4 – 5 cm of the outer wall of the capillary outlet is coated with silver conductive paint (Ag).14 This coating enables electrical contact between the electrolyte inside the capillary and the ground. By varying the thickness of the silver paint, the outer diameter of the capillary can be adjusted to tightly fit within the central tube of the nebulizer. This design creates a vacuum through rapid removal of the nebulizing gas, which facilitates passive liquid aspiration into the nebulizer, eliminating the need for an external sheath liquid. This configuration offers simplicity and ease of operation. However, this method has several limitations. It may induce peak broadening due to laminar flow, where the central portion of the liquid moves faster than the outer layers. Additionally, Joule heating can occur due to reduced resistance. If the analyte matrix contains high concentrations of interfering substances, undesired complexes may form with components of the conductive coating, potentially leading to contamination of the CE effluent and degradation of the coating.14 In the latter approach, a platinum wire (Pt wire) is bent and inserted into the outlet of the separation capillary, making direct contact with the electrolyte for electrical conduction.20 The other end of the Pt wire is routed back through the septum to the CE inlet and connected to the power supply unit, forming a closed electrical circuit. Like the previous method, this design eliminates the need for a sheath liquid flow. Nevertheless, this configuration also presents challenges. The 25 μm-diameter Pt wire may move freely within the capillary, potentially leading to mechanical instability. Moreover, portions of the capillary exposed to ambient air between the CE instrument and the ICP-MS interface are not thermally regulated, making them susceptible to Joule heating caused by the high-voltage CE process and thermal effects from the ICP source. These conditions can compromise run-to-run reproducibility due to temperature fluctuations.20
In CE, the sample injection volume is typically in the nanoliter (nL) range. To minimize sample loss and ensure efficient transfer to the plasma, it is essential to use low-flow rate, high-efficiency nebulizers. Several commercially available micro-flow nebulizers have been developed specifically for CE–ICP-MS coupling to address this requirement. These commercial nebulizers are often configured using simple connective components, similar to those used in HPLC systems, to integrate the separation capillary outlet, sheathliquid solution, platinum electrode, and nebulizing gas into a functional interface. These systems operate at flow rates that are nearly identical to those of the CE effluent, qualifying them as low-flow rate nebulizers. Due to their reduced capillary diameters and mixing volumes, they are commonly referred to as microconcentric nebulizers (MCNs). These nebulizers utilize either self-aspiration mechanisms or syringe-pumped sheath-liquid flow, which effectively eliminates suctionrelated issues at the capillary outlet. As a result, the high separation efficiency inherent to CE can be preserved, while also reducing dead volume and analyte dilution, thereby improving detection sensitivity. Unlike laboratory-made interfaces, which often require manual alignment and assembly, these commercial nebulizers allow for precise and reproducible positioning of the separation capillary within the center of the nebulizer. This reduces dependency on operator skill and improves ease of use. Furthermore, these systems offer enhanced connection stability, as evidenced by lower relative standard deviation (RSD) values in analytical results.21 Representative examples of such devices include the CEI-10022 (CETAC Technologies, Omaha, NE, USA) and the MiraMist CE (Burgener Research Inc., Mississauga, Ontario, Canada), as shown in Fig. 4. Other notable models are the MCN-100 and the PFA-100 (Elemental Scientific Inc., Omaha, USA).23
Since the first publication on CE-ICP-MS in 1995, research over the following two decades primarily focused on the development of coupling interfaces, improvement of detection sensitivity, and elemental/ speciation analysis across various sample types. Biologically relevant concentrations of trace elements typically range from nanomolar to micromolar levels (i.e., tens of ppb to several ppm),12 and numerous review articles confirm that the limits of detection (LODs) achieved by CE-ICP-MS—typically in the low to mid-ppb range—are sufficiently sensitive to meet these analytical demands.1-3,24 A keyword search for “CE-ICP-MS” or “capillary electrophoresis inductively coupled plasma mass spectrometry” on ScienceDirect from 2016 to 2025 yields approximately 1,000 research articles, indicating sustained and diverse interest in this technique. In the past decade, advancements such as commercialized interfaces have significantly broadened the scope of CE-ICPMS applications. Recent studies have expanded into more complex matrices and sophisticated analytical targets, including nanoparticle characterization,25 gene expression analysis,26 identification of metal-binding ligands such as zinc complexes,27 micronutrient monitoring in cyanobacterial culture media,28 and intracellular biotransformation pathway tracking.29 Table 1 summarizes the target analytes, nebulizers, and CE buffer systems used in representative recent CE-ICP-MS studies. It is notable that commercial nebulizers have become increasingly prevalent. Moreover, the application modes of CE have expanded—for example, coupling capillary gel electrophoresis with ICP-MS has enabled the size-based separation of DNA fragments, offering a cost-effective and time-saving alternative to conventional qPCR assays. These recent advancements, along with improvements in interface robustness and ease of use, are driving the continued expansion of CE-ICP-MS in diverse fields of elemental and speciation analysis.30
CE-ICP-MS has emerged as a powerful and versatile analytical platform for elemental and chemical speciation analysis. Since its inception in 1995, substantial advancements have been made in addressing the technical challenges associated with interface design and optimization of their interface parameters. Early research efforts were predominantly focused on improving coupling efficiency and enhancing detection sensitivity—necessitated by the inherent limitations of CE, including its requirement for small sample volumes and the complexity of establishing a stable electrical connection at the capillary outlet. The development and commercialization of low-flow nebulizers and more robust interface configurations have significantly improved the reliability, reproducibility, and overall usability of CE-ICP-MS systems. As a result, the scope of CE-ICP-MS applications has broadened considerably in recent years, extending beyond traditional elemental analysis to encompass increasingly complex biological and environmental matrices. The technique has been effectively applied in areas such as nanoparticle characterization, gene expression profiling, identification of metal-ligand complexes, monitoring of trace nutrients in microbial cultures, and the study of intracellular metal trafficking at the single-cell level. Moreover, the integration of capillary gel electrophoresis (CGE) with ICP-MS has demonstrated significant potential for the highresolution separation of biomolecules such as DNA, offering notable advantages over conventional molecular biology techniques in terms of costeffectiveness, analytical throughput, and sensitivity. Given its superior separation efficiency, excellent detection limits, minimal sample consumption, and wide-ranging applicability, CE-ICP-MS is wellpositioned to become an indispensable tool in trace element and speciation analysis.
S. S. Kannamkumarath, K. Wrobel, K. Wrobel, C. B'Hymer, and J. A. Caruso, J. Chromatogr. A, 975(2), 245-266 (2002). https://doi.org/10.1016/S0021-9673(02)01218-9 https://doi.org/10.1016/S0021-9673(02)01218-9
G. Álvarez-Llamas, M. a. d. R. Fernández de laCampa, and A. Sanz-Medel, TrAC, Trends Anal. Chem., 24(1), 28-36 (2005). https://doi.org/10.1016/j.trac.2004.09.007 https://doi.org/10.1016/j.trac.2004.09.007
B. Michalke, Eelectrophoresis, 26(7-8), 1584-1597 (2005). https://doi.org/10.1002/elps.200410314 https://doi.org/10.1002/elps.200410314
N. Muhammad, I. Hussain, A. Ali, L. Noureen, Q. He, Q. Subhani, N. A. Khan, H. Cui, and Y. Zhu, J. Chromatogr. A, 1734, 465311 (2024). https://doi.org/10.1016/j.chroma.2024.465311 https://doi.org/10.1016/j.chroma.2024.465311
A. R. Timerbaev, Chem. Rev., 113(1), 778-812 (2013). https://doi.org/10.1021/cr300199v https://doi.org/10.1021/cr300199v
H. Holtkamp, G. Grabmann, and C. G. Hartinger, Electrophoresis, 37(7-8), 959-972 (2016). https://doi.org/10.1002/elps.201500502 https://doi.org/10.1002/elps.201500502
J. W. Jorgenson and K. D. Lukacs, Anal. Chem., 53(8), 1298-1302 (1981). https://doi.org/10.1021/ac00231a037 https://doi.org/10.1021/ac00231a037
R. Thomas, Spectroscopy, 4, 38-42 (2001). https://doi.org/10.1016/S0341-8162(00)00132-6
R. Thomas, Spectroscopy, 5, 56-60 (2001). https://doi.org/10.1016/S0262-1762(01)80220-0
R. Thomas, Spectroscopy, 7, 26-34 (2001). https://doi.org/10.1097/00007632-200110010-00004
S. Wilschefski and M. Baxter, Clin Biochem Rev., 40, 115-133 (2019). https://doi.org/10.33176/AACB-19-00024 https://doi.org/10.33176/AACB-19-00024
R. Thomas, Spectroscopy, 10, 44-48 (2001). https://doi.org/10.1049/esej:20010202
J. W. Olesik, J. A. Kinzer, and S. V. Olesik, Anal. Chem., 67(1), 1-12 (1995). https://doi.org/10.1021/ac00097a003 https://doi.org/10.1021/ac00097a003
Y. Liu, V. Lopez-Avila, J. J. Zhu, D. Wiederin, and W. F. Beckert, Anal. Chem., 67(13), 2020-2025 (1995). https://doi.org/10.1021/ac00109a019 https://doi.org/10.1021/ac00109a019
J. A. Kinzer, J. W. Olesik, and S. V. Olesik, Anal. Chem., 68(18), 3250-3257 (1996). https://doi.org/10.1021/ac951143k https://doi.org/10.1021/ac951143k
Y. Li, J.-M. Liu, Y.-L. Xia, Y. Jiang, and X.-P. Yan, Electrophoresis, 29(22), 4568-4574 (2008). https://doi.org/10.1002/elps.200800309 https://doi.org/10.1002/elps.200800309
V. Majidi and N. J. Miller-Ihli, Analyst, 123(5), 803-808 (1998). https://doi.org/10.1039/A707770J https://doi.org/10.1039/A707770J
Y. Y. Chan and W. T. Chan, J. Chromatogr. A, 853(1), 141-149 (1999). https://doi.org/10.1016/S0021-9673(99)00713-X https://doi.org/10.1016/S0021-9673(99)00713-X
V. Majidi, Microchem. J., 66(1), 3-16 (2000). https://doi.org/10.1016/S0026-265X(00)00062-X https://doi.org/10.1016/S0026-265X(00)00062-
J. E. Sonke and V. J. M. Salters, J. Chromatogr. A, 1159(1), 63-74 (2007). https://doi.org/10.1016/j.chroma.2007.05.055 https://doi.org/10.1016/j.chroma.2007.05.055
D. Schaumlöffel and A. Prange, Fresenius' J. Anal. Chem., 364(5), 452-456 (1999). https://doi.org/10.1007/s002160051366 https://doi.org/10.1007/s002160051366
A. Prange and D. Pröfrock, Anal. Bioanal. Chem., 383(3), 372-389 (2005). https://doi.org/10.1007/s00216-005-3420-0 https://doi.org/10.1007/s00216-005-3420-0
H. Qu, T. K. Mudalige, and S. W. Linder, J. Chromatogr. A, 1429, 348-353 (2016). https://doi.org/10.1016/j.chroma.2015.12.033 https://doi.org/10.1016/j.chroma.2015.12.033
A. Fernández Asensio, T. Iglesias, A. Cotarelo, M. Espina, E. Blanco-González, L. M. Sierra, and M. Montes-Bayón, Anal. Chim. Acta, 1023, 64-73 (2018). https://doi.org/10.1016/j.aca.2018.03.047 https://doi.org/10.1016/j.aca.2018.03.047
L. Ruzik and P. Kwiatkowski, Talanta, 183, 102-107 (2018). https://doi.org/10.1016/j.talanta.2018.02.040 https://doi.org/10.1016/j.talanta.2018.02.040
N. Melicherová, T. Vaculovič, R. Kočí, M. Trtílek, J. Lavická, and F. Foret, Anal. Biochem., 694, 115630 (2024). https://doi.org/10.1016/j.ab.2024.115630 https://doi.org/10.1016/j.ab.2024.115630
X. Men, C.-X. Wu, X. Zhang, X. Wei, W.-Q. Ye, M.-L. Chen, T. Yang, Z.-R. Xu, and J.-H. Wang, Anal. Chim. Acta, 1226, 340268 (2022). https://doi.org/10.1016/j.aca.2022.340268 https://doi.org/10.1016/j.aca.2022.340268
H. U. Holtkamp, S. J. Morrow, M. Kubanik, and C. G. Hartinger, J. Chromatogr. A, 1561, 76-82 (2018). https://doi.org/10.1016/j.chroma.2018.05.036 https://doi.org/10.1016/j.chroma.2018.05.036